Future theranostic strategies: emerging ovarian cancer biomarkers to bridge the gap between diagnosis and treatment
- PMID: 40836974
- PMCID: PMC12363270
- DOI: 10.3389/fddev.2024.1339936
Future theranostic strategies: emerging ovarian cancer biomarkers to bridge the gap between diagnosis and treatment
Abstract
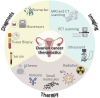
Ovarian cancers are a complex and heterogenic group of malignancies that are difficult to detect, diagnose and treat. Fortunately, considerable knowledge of ovarian cancer specific biomarkers has been generated, that is pertinent to the development of novel theranostic platforms by combining therapies and diagnostics. Genomic and proteomic data has been invaluable in providing critical biomolecular targets for ovarian cancer theranostic approaches. Exploitation of the wealth of biomarker research that has been conducted offers viable targets as beacons for ovarian cancer detection, diagnosis, and therapeutic targeting. These markers can be used in theranostics, a treatment strategy that combines therapy and diagnostics and is common in nuclear medicine, where radionuclides are used for both diagnosis and treatment. The development of theranostics has taken substantial focus in recent years in the battle against ovarian cancer. Yet to date only one theranostic technology has emerged in clinical practice. However, given the wealth of ovarian cancer biomarkers the field is poised to see the emergence of revolutionary disease treatment and monitoring outcomes through their incorporation into the development of theranostic strategies. The future of ovarian cancer treatment is set to enable precise diagnosis, targeted treatment, and vigilant monitoring. This review aims to assess the status of ovarian cancer diagnostic tools and biomarkers in practice, clinical development, or pre-clinical development, highlighting newly emerging theranostic applications.
Keywords: biomarkers; diagnostics; nanomedicine; ovarian cancer; theragnostics; theranostics; therapeutics; tumor.
Copyright © 2024 Rajapaksha, Khetan, Johnson, Blencowe, Garg, Albrecht and Gillam.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
Similar articles
-
Prescription of Controlled Substances: Benefits and Risks.2025 Jul 6. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2025 Jul 6. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 30726003 Free Books & Documents.
-
Management of urinary stones by experts in stone disease (ESD 2025).Arch Ital Urol Androl. 2025 Jun 30;97(2):14085. doi: 10.4081/aiua.2025.14085. Epub 2025 Jun 30. Arch Ital Urol Androl. 2025. PMID: 40583613 Review.
-
A rapid and systematic review of the clinical effectiveness and cost-effectiveness of topotecan for ovarian cancer.Health Technol Assess. 2001;5(28):1-110. doi: 10.3310/hta5280. Health Technol Assess. 2001. PMID: 11701100
-
The Black Book of Psychotropic Dosing and Monitoring.Psychopharmacol Bull. 2024 Jul 8;54(3):8-59. Psychopharmacol Bull. 2024. PMID: 38993656 Free PMC article. Review.
-
Short-Term Memory Impairment.2024 Jun 8. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2024 Jun 8. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 31424720 Free Books & Documents.
References
-
- A2 Biotherapeutics Inc (2023). A seamless phase 1/2 study to evaluate the safety and efficacy of A2B694, an autologous logic-gated TmodTM CAR T, in heterozygous HLA-A*02 adults with recurrent unresectable. Available from: https://clinicaltrials.gov/study/NCT06051695.
-
- AGO Research GmbH (2023). “A randomized phase II trial of mirvetuximab soravtansine (IMGN853),” in Folate receptor alpha (FRα) high recurrent ovarian cancer eligible for platinum-based chemotherapy. Available from: https://clinicaltrials.gov/study/NCT04274426.
-
- Agyemang A. F., Lele S. (2022). “The use of immunotherapy for treatment of gynecologic malignancies,” in Ovarian cancer. Editor Lele S. (Brisbane (AU): Exon Publications; ). - PubMed
-
- Allife Medical Science and Technology Co., Ltd (2019). Clinical study on the safety and efficacy of anti-mesothelin car NK cells with epithelial ovarian cancer. Available from: https://clinicaltrials.gov/study/NCT03692637.
Publication types
LinkOut - more resources
Full Text Sources
Miscellaneous