Stem/progenitor cell dynamics during salivary gland development and regeneration demonstrated by the double pulse-chase paradigm
- PMID: 40837863
- PMCID: PMC12362022
- DOI: 10.1016/j.reth.2025.08.007
Stem/progenitor cell dynamics during salivary gland development and regeneration demonstrated by the double pulse-chase paradigm
Abstract
Introduction: Active and quiescent stem cells coexist in hair follicles and intestinal crypts; however, their localization and differentiation potential in the salivary dynamics are unknown. This study aimed to clarify the cellular dynamics that occur during salivary gland development and regeneration in the duct ligation model with a focus on the role of label retaining cells (LRCs), presumably quiescent stem cells, in these processes.
Methods: Doxycycline-inducible TetOP-histone 2B (H2B)-green fluorescent protein (GFP) transgenic mice [GFP expression was induced during embryonic day 15 (E15)-postnatal day 7 (P7)] followed by EdU (5-ethynyl-2'-deoxyuridine) administration at P10-14 to chase the LRCs during development. In addition, LRCs were labeled with GFP immediately before salivary gland duct ligation and EdU was administered after the ligation was released to chase the LRCs with GFP and EdU during tissue repair.
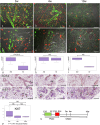
Results and conclusions: During development, GFP (+) EdU (-) LRCs were abundant in striated duct cells (SDCs) and GFP (+) EdU (+) LRCs were primarily localized to the intercalated duct cells (IDCs) at P21. Labeling of the GFP (+) LRCs faded as well as the EdU (+) LRCs at P70. During tissue repair, GFP (+) EdU (+) LRCs were colocalized in the IDCs and myoepithelium cells (MECs), whereas the GFP (+) EdU (+) acinar cells (ACs) appeared over time. These results suggest that salivary gland quiescent stem/progenitor cells are present in the IDCs during development and that quiescent stem/progenitor cells in the IDCs and MECs differentiate into ACs during tissue repair.
Keywords: Adult stem cell; Growth and development; Regeneration; Salivary glands; Transgenic mice.
© 2025 The Author(s).
Conflict of interest statement
The authors declare no conflicts of interest related to this study.
Figures











References
-
- Yeh C.K., Johnson D.A., Dodds M.W. Impact of aging on human salivary gland function: a community-based study. Aging (Milano) 1998;10:421–428. - PubMed
-
- Choi J.S., Park I.S., Kim S.K., Lim J.Y., Kim Y.M. Analysis of age-related changes in the functional morphologies of salivary glands in mice. Arch Oral Biol. 2013;58:1635–1642. - PubMed
-
- Enoki N., Kiyoshima T., Sakai T., Kobayashi I., Takahashi K., Terada Y., Sakai H. Age-dependent changes in cell proliferation and cell death in the periodontal tissue and the submandibular gland in mice: a comparison with other tissues and organs. J Mol Histol. 2007;38:321–332. - PubMed
-
- Lan X., Chan J.Y.K., Pu J.J., Qiao W., Pang S., Yang W.F., Wong K.C.W., Kwong D.L.W., Su Y.X. Saliva electrolyte analysis and xerostomia-related quality of life in nasopharyngeal carcinoma patients following intensity-modulated radiation therapy. Radiother Oncol. 2020;150:97–103. - PubMed
LinkOut - more resources
Full Text Sources

