Safety and efficacy of MCO-010 optogenetic therapy in patients with Stargardt disease in USA (STARLIGHT): an open-label multi-center Ph2 trial
- PMID: 40838197
- PMCID: PMC12362400
- DOI: 10.1016/j.eclinm.2025.103430
Safety and efficacy of MCO-010 optogenetic therapy in patients with Stargardt disease in USA (STARLIGHT): an open-label multi-center Ph2 trial
Abstract
Background: Stargardt disease (SD) is an inherited degenerative retinal disease affecting rod and cone photoreceptors and retinal pigment epithelial (RPE) cells, leading to severe and irreversible vision loss. Optogenetics is a promising approach to restoring vision by photosensitizing spared healthy retinal neurons.
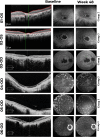
Methods: The STARLIGHT phase 2 open-label study (NCT05417126) was conducted over 48-weeks at two US sites to assess the safety and efficacy of MCO-010 administered via a single intravitreal injection in the worse-seeing eye of SD patients. The study began on July 5, 2022, and was completed on September 28, 2023. MCO-010 targets bipolar cells rather than retinal ganglion cells (RGCs) to utilize more abundant cells that respond to ambient light while preserving natural visual processing pathways after treatment. Six adults (mean age 50 years, range 32-71 years, four males and two females) received 1.2E11 genome copies (gc)/eye of MCO-010. The primary outcome was the incidence, nature, severity of treatment-emergent adverse events (TEAEs), serious adverse events (SAEs), intraocular inflammation, retinal thickness, best-corrected visual acuity (BCVA), and lesion size. Secondary endpoints included change in BCVA, vision-guided mobility, and shape determination accuracy. Visual field perimetry and Michigan Retinal Degeneration Questionnaire (MRDQ) were exploratory endpoints.
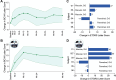
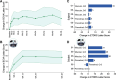
Findings: All six participants had at least one ocular TEAE; non-ocular TEAEs occurred in three participants. The most common TEAEs were conjunctival hemorrhage, ocular hypertension, and vitreous cells (two subjects). There were no deaths, hospitalization, loss of eye, retinal detachment, endophthalmitis, or TEAEs leading to study discontinuation. The BCVA (mean ± SD) of the six treated eyes at baseline and 48-week follow-up was 22.8 ± 9.87 (range 9-35), and 28.3 ± 13.28 (range 4-42) ETDRS letters, respectively. The BCVA change from baseline was 7.2 ± 11.74, 4.2 ± 14.81, and 5.5 ± 12.29 at 12, 24, and 48 weeks, respectively. With a wearable magnifier (low-vision glasses), the BCVA change was 17.8 ± 13.35, 15.7 ± 17.37, 13.3 ± 21.37 at 12, 24, and 48 weeks, respectively. The improvement in mean defect in visual field perimetry was 1.02 ± 3.54, 2.47 ± 5.00, and 2.63 ± 5.26 dB at 12, 24, and 48 weeks, respectively. Specific improvements were noted in reading, and color, and contrast domains of the MRDQ.
Interpretation: MCO-010 optogenetic phase 2 results support further investigation for treatment SD. To our knowledge, this is the first report of improving on(eye)-chart vision of SD participants utilizing optogenetics.
Funding: Nanoscope Therapeutics Inc.
Keywords: Multi-characteristic opsin; Optogenetics; Retinal degenerative disease; Stargardt macular degeneration; Vision restoration.
© 2025 The Author(s).
Conflict of interest statement
BLL and VHG received research funding from Nanoscope Therapeutics for site participation in the STARLIGHT trial. BLL and NG received research funding from Ascidian Therapeutics, Belite Bio, Ocugen, and SpliceBio for site participation in clinical trials for Stargardt disease. BLL and NGG is also a paid consultant for SpliceBio. The authors VHG, SHC, SB, MC, SK, AA, JC, HL, NC, MVT, NAS, ACH, SBB, and SKM have an equity interest in Nanoscope Therapeutics Inc. BLL, JK, JPS, SHT, ACH, VBM are paid consultants of Nanoscope. JK is a paid consultant for Kubota Vision, and Belite Bio. DB has an equity interest in Ocugen and is a paid consultant for Ocugen and Ray Therapeutics, developing gene therapies for Stargardt disease. SKM has issued and pending patents on the MCO technology platform.
Figures


References
-
- Kaplan J., Gerber S., Larget-Piet D., et al. A gene for Stargardt’s disease (fundus flavimaculatus) maps to the short arm of chromosome 1. Nat Genet. 1993;5:308–311. - PubMed
-
- Allikmets R., Singh N., Sun H., et al. A photoreceptor cell-specific ATP-Binding transporter gene (ABCR) is mutated in recessive Starqardt macular dystrophy. Nat Genet. 1997;15:236–246. - PubMed
-
- Cideciyan A.V., Aleman T.S., Swider M., et al. Mutations in ABCA4 result in accumulation of lipofuscin before slowing of the retinoid cycle: a reappraisal of the human disease sequence. Hum Mol Genet. 2004;13:525–534. - PubMed
-
- Oh K.T., Weleber R.G., Stone E.M., Oh D.M., Rosenow J., Billingslea A.M. Electroretinographic findings in patients with Stargardt disease and fundus flavimaculatus. Retina. 2004;24:920–928. - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

