Using sputum and tongue swab specimens for in-home point-of-care targeted universal testing for tuberculosis of household contacts: an acceptability and feasibility analysis
- PMID: 40840972
- PMCID: PMC12374641
- DOI: 10.1136/bmjgh-2024-018131
Using sputum and tongue swab specimens for in-home point-of-care targeted universal testing for tuberculosis of household contacts: an acceptability and feasibility analysis
Abstract
Introduction: Effective strategies are essential for early tuberculosis (TB) detection. Reliance on passive case detection, symptom screening and collection of sputum results in delayed or undiagnosed TB, contributing to on-going TB transmission. This study assessed the acceptability of in-home targeted universal TB testing (TUTT) using GeneXpert MTB/RIF Ultra at point-of-care (POC) during household contact investigations (HCIs) and the feasibility of using sputum and tongue swab specimens.
Methods: The TB Home Study sought to evaluate the predictive value of different specimen types for use as a household-level triage test for TB. Household contacts of people with TB residing in the Buffalo City Metro Health District (Eastern Cape Province, South Africa) who received in-home POC TUTT through the TB Home Study were asked to complete a post-test acceptability survey. The survey assessed the level of comfort, confidence in the test results and perceived appropriateness of in-home POC TUTT. A feasibility framework was used to assess the feasibility of using sputum and tongue swab specimens for testing.
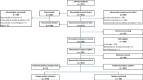
Results: Of the 325 eligible household contacts, 281/325 (86.5%) provided consent. Of those contacts, 278/281 (98.9%) provided a tongue swab, and 50/281 (17.8%) could expectorate sputum. All specimens were successfully prepared for immediate in-home testing. Of the 172 tongue swab-based tests performed, 169 (98.3%) produced a valid result, whereas 47 of 49 (95.9%) sputum-based tests had a valid result. An immediate tongue swab-based test result was available for 274/278 (98.6%) clients compared with 47/49 (95.9%) sputum-based test results. The mean in-home POC TUTT acceptability score (5=highly acceptable) was 4.2/5 (SD=0.4).
Conclusion: In-homePOC TUTT using sputum and tongue swab specimens was highly acceptable and feasible. Tongue swabs greatly increased the testing rates owing to the high sample collection yield. Combining sputum and tongue swabs for in-home POC testing offers a promising strategy to improve TB case detection and reduce diagnostic delays.
Keywords: Delivery of Health Care; Health Services Accessibility; Tuberculosis.
© Author(s) (or their employer(s)) 2025. Re-use permitted under CC BY-NC. No commercial re-use. See rights and permissions. Published by BMJ Group.
Conflict of interest statement
Competing interests: None declared.
Figures
Update of
-
USING SPUTUM AND TONGUE SWAB SPECIMENS FOR IN-HOME POINT-OF-CARE TARGETED UNIVERSAL TESTING FOR TB OF HOUSEHOLD CONTACTS: AN ACCEPTABILITY AND FEASIBILITY ANALYSIS.medRxiv [Preprint]. 2024 Nov 2:2024.11.01.24316570. doi: 10.1101/2024.11.01.24316570. medRxiv. 2024. Update in: BMJ Glob Health. 2025 Aug 21;10(8):e018131. doi: 10.1136/bmjgh-2024-018131. PMID: 39574838 Free PMC article. Updated. Preprint.
References
-
- Global tuberculosis report 2024. 1st. Geneva: World Health Organization; 2024. p. 1. edn.
-
- The South African National Department of Health TB-recovery-plan-2.0, April-2023-March-2024. [14-Oct-2024]. https://tbthinktank.org/wp-content/uploads/2023/09/TB-Recovery-Plan-2.0-... Available. Accessed.
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical