Financial toxicity in lower urinary tract symptoms amongst men
- PMID: 40841616
- PMCID: PMC12372289
- DOI: 10.1186/s12894-025-01895-4
Financial toxicity in lower urinary tract symptoms amongst men
Abstract
Background: Financial toxicity (FT), first reported in oncologic patients and generally defined as harm to patients caused by the cost of treatment, is less well described in non-malignant urology. In chronic conditions related to lower urinary tract symptoms (LUTS), such as BPH, treatment costs may result in a significant substantial burden. The goal of this study was to characterize the association between FT and LUTS.
Methods: A cross-sectional web-based survey was administered to a random sample of adult men through a national registry of volunteers (ResearchMatch). Disease-specific information, validated symptom scores, and an 11-item measure of LUTS-related financial toxicity were used to characterize participants. Multivariable logistic regression was performed to identify predictors of financial toxicity.
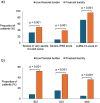
Results: A total of 294 respondents with a self-reported history of BPH-associated LUTS were included, 41% of whom met the criteria for financial toxicity. Men with FT had worse LUTS symptom scores across all measures (p < 0.001). The presence of stress, urge, or mixed incontinence was significantly higher in men with FT (52% vs. 8%, 46% vs. 15%, and 36% vs. 5% respectively; p < 0.001). Men with FT spent more out-of-pocket on incontinence products than those without FT (p < 0.001). On multivariable analysis, mixed incontinence was a predictor of FT (OR 3.233, 95% CI 1.15–9.084).
Conclusion: Two of five men with LUTS met the criteria for financial toxicity. These men had worse urinary symptom scores, higher rates of all types of incontinence, and higher out-of-pocket costs for incontinence products.
Supplementary Information: The online version contains supplementary material available at 10.1186/s12894-025-01895-4.
Keywords: Financial stress; Health expenditures; Lower urinary tract symptoms; Prostatic hyperplasia; Urinary incontinence.
Conflict of interest statement
Declarations. Ethics approval and consent to participate: All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. The study was IRB-approved (IRB #23-38991) approved by the Bioethics Committee of the University of California (UCSF) Medical School. Consent for publication: Participants were informed that completing the survey implied consent to participate in the study. No personally identifiable information was collected, and responses were anonymized to protect participants’ privacy. Given the anonymous nature of the survey and the minimal risk involved, formal written consent was not required, in accordance with institutional and ethical guidelines. Competing interests: The authors declare no competing interests.
Figures
References
-
- Berry SJ, Coffey DS, Walsh PC, Ewing LL. The development of human benign prostatic hyperplasia with age. J Urol. 1984;132:474–9. 10.1016/s0022-5347(17)49698-4. - PubMed
-
- Welch G, Weinger K, Barry MJ. Quality-of-life impact of lower urinary tract symptom severity: results from the health professionals Follow-up study. Urology. 2002;59:245–50. 10.1016/S0090-4295(01)01506-0. - PubMed
-
- Irwin DE, Milsom I, Hunskaar S, Reilly K, Kopp Z, Herschorn S, et al. Population-based survey of urinary incontinence, overactive bladder, and other lower urinary tract symptoms in five countries: results of the EPIC study. Eur Urol. 2006;50:1306–14. 10.1016/j.eururo.2006.09.019. discussion 1314–1315. - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

