Targeting cancer-associated fibroblast-driven LIF/LIFR axis improves the therapeutic efficacy of gemcitabine and nab-paclitaxel in pancreatic cancer
- PMID: 40846751
- PMCID: PMC12373761
- DOI: 10.1038/s41698-025-01046-w
Targeting cancer-associated fibroblast-driven LIF/LIFR axis improves the therapeutic efficacy of gemcitabine and nab-paclitaxel in pancreatic cancer
Abstract
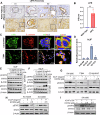
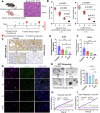
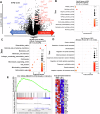
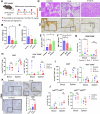
Pancreatic ductal adenocarcinoma (PDAC) is inherently therapy resistant due to cancer cell-stroma crosstalk across several signaling pathways. Among these, the LIF/LIFR axis has been implicated in cancer cell and cancer-associated fibroblast (CAF) crosstalk. We evaluated the efficacy of EC359, a competitive inhibitor of LIFR, in combination with gemcitabine. EC359 reduced tumor burden by 90% compared to controls and by 55% compared to gemcitabine alone in cancer cell and CAFs co-implannation model. The RNA-seq analysis revealed a significant alteration in extracellular matrix components, stemness, microtubule assembly, and immune response, suggesting simultaneous targeting of cancer cell-intrinsic and stroma-mediated mechanisms by EC359. In autochthonous murine model of PDAC, EC359 enhanced the therapeutic efficacy of gemcitabine and nab-paclitaxel, accompanied by an increase in dendritic cells but a reduction in T-regulatory cells. Thus, EC359 reduces PDAC cell stemness, stabilizes microtubule assembly, and reduces the immunosuppressive microenvironment to improve the efficacy of standard-of-care in PDAC.
© 2025. The Author(s).
Conflict of interest statement
Competing interests: S.K.B. is one of the founders of Sanguine Diagnostics and Therapeutics, Inc. H.N., S.B. and A.G. are employees of Evestra and holders of a patent on EC359. The other authors declare no competing interests.
Figures

References
-
- Lohr, M. et al. Transforming growth factor-β1 induces desmoplasia in an experimental model of human pancreatic carcinoma. Cancer Res.61, 550–555 (2001). - PubMed
-
- Giannoni, E. et al. Reciprocal activation of prostate cancer cells and cancer-associated fibroblasts stimulates epithelial-mesenchymal transition and cancer stemness. Cancer Res.70, 6945–6956 (2010). - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

