Interplay of genetic predisposition, plasma metabolome and Mediterranean diet in dementia risk and cognitive function
- PMID: 40855194
- PMCID: PMC12618253
- DOI: 10.1038/s41591-025-03891-5
Interplay of genetic predisposition, plasma metabolome and Mediterranean diet in dementia risk and cognitive function
Erratum in
-
Author Correction: Interplay of genetic predisposition, plasma metabolome and Mediterranean diet in dementia risk and cognitive function.Nat Med. 2025 Nov;31(11):3930. doi: 10.1038/s41591-025-04025-7. Nat Med. 2025. PMID: 41039117 Free PMC article. No abstract available.
Abstract
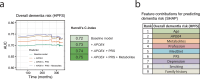
Alzheimer's disease (AD) and AD-related dementias (AD/ADRD) have a substantial genetic basis, with APOE4 homozygotes increasingly recognized as a distinct genetic subtype. To identify genotype-specific metabolic pathways and modifiable risk factors, we integrated genetic, plasma metabolomic and dietary data from 4,215 women and 1,490 men in prospective cohorts. Here we show that the associations of 57 metabolites with dementia risk varied by APOE4 genotype or other AD/ADRD risk variants. For example, cholesteryl esters and sphingomyelins were most strongly associated with increased dementia risk in APOE4 homozygotes, whereas inverse associations with glycerides were specific to this genotype. Dimethylguanidino-valeric acid was more strongly associated with dementia risk among carriers of the rs2154481-C allele (APP). Adherence to the Mediterranean diet more effectively modulated dementia-related metabolites in APOE4 homozygotes, suggesting targeted prevention strategies. Incorporating metabolomic data modestly improved dementia risk prediction, particularly during early follow-up. Mendelian randomization analysis identified 19 putative causal relationships between metabolites and cognitive outcomes, including protective effects of 4-guanidinobutanoate, carotenoids and N6-carbamoylthreonyladenosine. These findings reveal genotype-dependent metabolic profiles of cognitive health and support precision nutrition approaches for ADRD prevention.
© 2025. This is a U.S. Government work and not under copyright protection in the US; foreign copyright protection may apply.
Conflict of interest statement
Competing interests: The authors declare no competing interests.
Figures













References
MeSH terms
Substances
Grants and funding
- U54AG089325/U.S. Department of Health & Human Services | NIH | National Institute on Aging (U.S. National Institute on Aging)
- P01 CA087969/CA/NCI NIH HHS/United States
- RF1AG083764/U.S. Department of Health & Human Services | NIH | National Institute on Aging (U.S. National Institute on Aging)
- U01 CA167552/CA/NCI NIH HHS/United States
- R01NR019992/U.S. Department of Health & Human Services | NIH | National Institute of Nursing Research (NINR)
- R01 NR019992/NR/NINR NIH HHS/United States
- P30 DK046200/DK/NIDDK NIH HHS/United States
- R00DK119412/U.S. Department of Health & Human Services | NIH | National Institute of Diabetes and Digestive and Kidney Diseases (National Institute of Diabetes & Digestive & Kidney Diseases)
- R01 AG077489/AG/NIA NIH HHS/United States
- R01 HL060712/HL/NHLBI NIH HHS/United States
- P30DK046200/U.S. Department of Health & Human Services | NIH | National Institute of Diabetes and Digestive and Kidney Diseases (National Institute of Diabetes & Digestive & Kidney Diseases)
- R01AG077489/U.S. Department of Health & Human Services | NIH | National Institute on Aging (U.S. National Institute on Aging)
- R00 DK119412/DK/NIDDK NIH HHS/United States
- R01 HL034594/HL/NHLBI NIH HHS/United States
- UM1 CA186107/CA/NCI NIH HHS/United States
- U54 AG089325/AG/NIA NIH HHS/United States
- RF1 AG083764/AG/NIA NIH HHS/United States
- R01 HL035464/HL/NHLBI NIH HHS/United States
- R01 HL088521/HL/NHLBI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Medical

