Model-informed exploration of the boundaries of safe aluminium exposure from allergen immunotherapy in children
- PMID: 40856306
- PMCID: PMC12379567
- DOI: 10.1111/pai.70181
Model-informed exploration of the boundaries of safe aluminium exposure from allergen immunotherapy in children
Abstract
Background: The safety of aluminium (Al) exposure from medicinal products for subcutaneous allergen immunotherapy (SCIT) is still under debate due to their administration in many doses over years. Especially for children, model-informed risk assessment is urgently needed in the absence of clinical study data.
Methods: We applied a physiologically-based toxicokinetic Al model for simulation of Al exposure from various SCIT scenarios in children (5 and 10 years) compared to adults (35 years) in addition to continuous Al exposure from dietary intake.
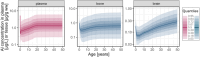
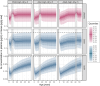
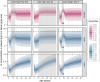
Results: Simulations of a worst-case Al exposure scenario (five-year SCIT; four-weekly injections; 1.25 mg Al per dose) in children reveal substantial, but subtoxic and transient increases in bone Al content. Predicted increases of Al levels in the brain appear negligible. In both of these storage organs, Al levels predicted at 50 years of age were only marginally affected by the previous SCIT treatment. Two exceptional SCIT scenarios, prolongation of treatment duration to 40 years and parallel treatment with two 1.25 mg Al-containing products, were associated with elevated bone Al levels above the normal adult range and close to a level where potential harm to bone metabolism cannot be excluded.
Conclusions: Simulations support the tolerability of single SCIT treatments in all age groups ≥5 years, also with respect to the long-term Al body burden. The Pharmacopoeial limit of 1.25 mg Al per dose could be seen as a conservative threshold also for the sum of doses from parallel treatments. The decision to prolong therapy with Al-containing SCIT products should be taken under careful consideration of Al accumulation.
Keywords: adjuvants; allergen immunotherapy; aluminium; computer simulation; immunologic; patient safety; toxicokinetics.
© 2025 The Author(s). Pediatric Allergy and Immunology published by European Academy of Allergy and Clinical Immunology and John Wiley & Sons Ltd.
Conflict of interest statement
The authors declare no conflict of interest in relation to this work.
Figures

Similar articles
-
Prescription of Controlled Substances: Benefits and Risks.2025 Jul 6. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2025 Jul 6. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 30726003 Free Books & Documents.
-
A systematic review and economic evaluation of subcutaneous and sublingual allergen immunotherapy in adults and children with seasonal allergic rhinitis.Health Technol Assess. 2013 Jul;17(27):vi, xi-xiv, 1-322. doi: 10.3310/hta17270. Health Technol Assess. 2013. PMID: 23827204 Free PMC article.
-
Allergen-specific oral immunotherapy for peanut allergy.Cochrane Database Syst Rev. 2012 Sep 12;2012(9):CD009014. doi: 10.1002/14651858.CD009014.pub2. Cochrane Database Syst Rev. 2012. PMID: 22972130 Free PMC article.
-
Specific allergen immunotherapy for the treatment of atopic eczema.Cochrane Database Syst Rev. 2016 Feb 12;2(2):CD008774. doi: 10.1002/14651858.CD008774.pub2. Cochrane Database Syst Rev. 2016. PMID: 26871981 Free PMC article.
-
One-strength dose escalation of house dust mite depot product for subcutaneous immunotherapy is safe and tolerable.Allergy. 2025 Mar;80(3):807-816. doi: 10.1111/all.16370. Epub 2024 Nov 14. Allergy. 2025. PMID: 39540587 Free PMC article. Clinical Trial.
References
-
- Klimek L, Brehler R, Hamelmann E, et al. Evolution of subcutaneous allergen immunotherapy (part 1): from first developments to mechanism‐driven therapy concepts. Allergo J Int. 2019;28:78‐95.
-
- European Medicines Agency . Allergen products development for immunotherapy and allergy diagnosis in moderate to low‐sized study populations – Scientific guideline 2025. https://www.ema.europa.eu/en/documents/scientific‐guideline/guideline‐al... Accessed July 24, 2025 - PMC - PubMed
-
- European Medicines Agency . PDCO standard paediatric investigation plan for allergen products for specific immunotherapy 2013. https://www.ema.europa.eu/en/documents/regulatory‐procedural‐guideline/e... Accessed July 24, 2025
-
- Roberts G, Pfaar O, Akdis CA, et al. EAACI guidelines on allergen immunotherapy: allergic rhinoconjunctivitis. Allergy. 2018;73(4):765‐798. - PubMed
-
- Sturm GJ, Varga EM, Roberts G, et al. EAACI guidelines on allergen immunotherapy: hymenoptera venom allergy. Allergy. 2018;73(4):744‐764. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

