Role of N6-methyladenosine methylation in transverse aortic constriction-induced cardiac fibrosis: insights from MeRIP-seq analysis
- PMID: 40856860
- PMCID: PMC12380991
- DOI: 10.1007/s11033-025-10940-2
Role of N6-methyladenosine methylation in transverse aortic constriction-induced cardiac fibrosis: insights from MeRIP-seq analysis
Abstract
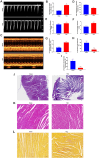
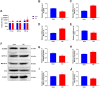
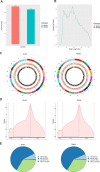
This study aimed to investigate the role and potential mechanisms of N6-methyladenosine (m6A) methylation in a mouse model of transverse aortic constriction (TAC)-induced cardiac fibrosis using MeRIP-seq. A TAC-induced cardiac fibrosis mouse model was established, and cardiac function and structural parameters were assessed by echocardiography four weeks post-surgery. The global m6A methylation levels in myocardial tissues were evaluated using Dot blot analysis, and the expression levels of m6A-modifying enzymes (METTL3, METTL14, ALKBH5, FTO) were detected by qPCR and Western blot. Additionally, methylated RNA immunoprecipitation sequencing (MeRIP-Seq) was performed to identify differentially methylated sites, followed by Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway enrichment analyses to determine the biological functions and signaling pathways of m6A-methylated genes. The results showed that TAC surgery successfully induced a cardiac fibrosis model in mice, as indicated by significantly reduced EF and LVEF and increased LVIDd and LVIDs. Moreover, m6A methylation levels were elevated in myocardial tissues of TAC mice, accompanied by upregulation of METTL3 and METTL14 expressions and downregulation of ALKBH5 and FTO expression. MeRIP-seq revealed that m6A peaks were primarily enriched in the 3' UTR regions, with 1,466 differentially methylated sites identified between TAC and sham groups, including 717 hypermethylated and 749 hypomethylated sites. Functional enrichment analyses showed that these differentially methylated genes were involved in various biological processes, including signal transduction, transcriptional regulation, and ion channel activity, and were associated with pathways such as type 2 diabetes mellitus, signaling pathways regulating pluripotency of stem cells, and insulin signaling. Thus, our findings suggested that m6A methylation played a significant role in TAC-induced cardiac fibrosis by regulating key genes involved in myocardial remodeling and functional impairment.
Keywords: Cardiac fibrosis; Methylated RNA immunoprecipitation sequencing; N6-methyladenosine; Transverse aortic constriction.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Competing interests: The authors declare no competing interests. Ethics approval and consent to participate: Ethical approval was provided by the Ethics Committee of Lanzhou University First Hospital, China (Approval: LDYYLL2025-122). Consent for publication: Not applicable.
Figures

Similar articles
-
m6A-related bioinformatics analysis and functional characterization reveals that METTL3-mediated NPC1L1 mRNA hypermethylation facilitates progression of atherosclerosis via inactivation of the MAPK pathway.Inflamm Res. 2023 Mar;72(3):429-442. doi: 10.1007/s00011-022-01681-0. Epub 2022 Dec 30. Inflamm Res. 2023. PMID: 36583755
-
N6-methyladenosine RNA landscape in the aged mouse hearts.Front Cardiovasc Med. 2025 Jun 18;12:1563364. doi: 10.3389/fcvm.2025.1563364. eCollection 2025. Front Cardiovasc Med. 2025. PMID: 40606020 Free PMC article.
-
Construction of RNA m6A profiles in liver tissue of mice in sepsis-induced liver injury based on m6A MeRIP-seq and RNA-seq.Eur J Med Res. 2025 Aug 8;30(1):723. doi: 10.1186/s40001-025-02985-7. Eur J Med Res. 2025. PMID: 40775729 Free PMC article.
-
Novel landscapes of N6-methyladenosine modification of mitochondrial oxidative stress in organ fibrosis.Eur J Pharmacol. 2025 Sep 15;1003:177888. doi: 10.1016/j.ejphar.2025.177888. Epub 2025 Jul 1. Eur J Pharmacol. 2025. PMID: 40609613 Review.
-
Mechanistic insights into the role of RNA demethylase ALKBH5 in malignant tumor therapy.J Transl Med. 2025 Aug 13;23(1):905. doi: 10.1186/s12967-025-06938-w. J Transl Med. 2025. PMID: 40804394 Free PMC article. Review.
References
-
- Fernández-Ruiz I (2019) Cardiac fibrosis research: two steps forward. Nat Rev Cardiol 16:515. 10.1038/s41569-019-0245-7 - PubMed
-
- Ravassa S et al (2023) Cardiac fibrosis in heart failure: focus on non-invasive diagnosis and emerging therapeutic strategies. Mol Aspects Med. 10.1016/j.mam.2023.101194 - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

