Intermittent fasting reprograms the brain proteome to prevent synaptic degeneration and cognitive impairment in vascular dementia
- PMID: 40860138
- PMCID: PMC12374579
- DOI: 10.7150/thno.119422
Intermittent fasting reprograms the brain proteome to prevent synaptic degeneration and cognitive impairment in vascular dementia
Abstract
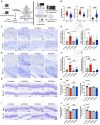
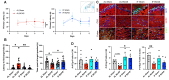
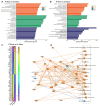
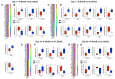
Rationale: Vascular dementia (VaD), driven by chronic cerebral hypoperfusion (CCH), leads to synaptic degeneration and cognitive decline, yet mechanisms linking vascular dysfunction to synaptic loss remain unclear. Intermittent fasting (IF) has emerged as a potential intervention, but its effects on synaptic integrity in VaD are unknown. This study aims to investigate the effects of IF against synaptic degeneration and cognitive impairment induced by CCH. Methods: Bilateral common carotid artery stenosis (BCAS) was employed to induce chronic CCH by placing 0.18 mm micro-coils around each common carotid artery in mice. To assess temporal differences, the coils remained in place for 1, 7, 14, or 30 days. IF was implemented for 16 hours daily over three months prior to BCAS induction. Cognitive impairment was evaluated using the Barnes maze test. White matter lesions (WMLs) and neuronal loss were assessed using Luxol fast blue and cresyl violet staining, respectively. Immunoblotting and immunohistochemistry were performed to quantify synaptic protein levels. Synaptic integrity was examined using transmission electron microscopy. Proteomic analysis of the hippocampus was conducted to investigate molecular adaptations to IF following CCH. Results: We demonstrate that a 16-hour IF regimen preserves cognitive function and synaptic density despite persistent hypoperfusion. Behavioral assays revealed that IF prevented spatial memory deficits in BCAS mice, while electron microscopy confirmed synaptic preservation without altering baseline architecture. Surprisingly, key synaptic protein levels remained unchanged, suggesting IF protects synaptic function rather than abundance. Proteomic profiling revealed dynamic hippocampal adaptations under IF, including upregulation of synaptic stabilizers, enhanced GABAergic signaling, and suppression of neuroinflammatory mediators. CCH induced microglial engulfment of synapses, suggesting a role in complement-mediated synaptic pruning. Temporal pathway analysis revealed IF's multi-phase neuroprotection: early synaptic reinforcement, mid-phase metabolic optimization, and late-phase suppression of chronic neuroinflammation. Conclusion: These findings establish IF as a potent modulator of synaptic resilience in VaD, acting through coordinated preservation of synaptic structure, inhibition of inflammatory synapse loss, and metabolic reprogramming. Our results highlight IF's potential as a non-pharmacological strategy to combat vascular cognitive impairment by targeting the synaptic vulnerability underlying dementia progression.
Keywords: cognitive impairment; intermittent fasting; neuronal death; synaptic loss; vascular dementia.
© The author(s).
Conflict of interest statement
Competing Interests: The authors have declared that no competing interest exists.
Figures



References
-
- van der Flier WM, Skoog I, Schneider JA, Pantoni L, Mok V, Chen CLH, Scheltens P. Vascular cognitive impairment. Nat Rev Dis Primers. 2018;4:18003. - PubMed
-
- Wang XX, Zhang B, Xia R, Jia QY. Inflammation, apoptosis and autophagy as critical players in vascular dementia. Eur Rev Med Pharmacol Sci. 2020;24:9601–14. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

