Reduced Brain Iron and Striatal Hyperdopaminergia in Schizophrenia: A Quantitative Susceptibility Mapping MRI and PET Study
- PMID: 40887951
- PMCID: PMC7618194
- DOI: 10.1176/appi.ajp.20240512
Reduced Brain Iron and Striatal Hyperdopaminergia in Schizophrenia: A Quantitative Susceptibility Mapping MRI and PET Study
Abstract
Objective: Neuroimaging studies have independently associated schizophrenia with low iron and elevated dopamine synthesis. While preclinical research demonstrates that midbrain iron deficiency leads to striatal hyperdopaminergia, this relationship has not been studied in schizophrenia. The authors conducted a case-control study to examine differences in tissue magnetic susceptibility, a marker of brain iron, and correlated these with striatal dopamine synthesis capacity.
Methods: Magnetic susceptibility in the substantia nigra and ventral tegmental area (SN-VTA) was measured using quantitative susceptibility mapping (QSM) MRI in 159 participants (control subjects, N=80; early-course schizophrenia, N=79, including patients who were antipsychotic-naïve or antipsychotic-free). Because magnetic susceptibility is increased by neuromelanin and reduced by myelin, neuromelanin-sensitive MRI (NM-MRI) and diffusion tensor imaging (DTI) were employed to investigate the influence of neuromelanin and myelin on the QSM findings in 99 participants (control subjects, N=38; schizophrenia patients, N=61). Dopamine synthesis capacity (Ki cer) was then assessed with [18F]-DOPA positron emission tomography in 40 people from the schizophrenia group to test whether low SN-VTA magnetic susceptibility was related to high striatal Ki cer.
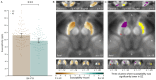
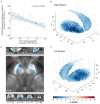
Results: SN-VTA magnetic susceptibility was lower in patients with schizophrenia than in control subjects (d=-0.66, 95% CI=-0.98, -0.34). This difference remained significant in analyses controlling for mean diffusivity (a DTI measure inversely correlating with myelin concentration) and NM-MRI contrast-to-noise ratios. SN-VTA magnetic susceptibility was significantly inversely correlated with striatal Ki cer, independent of mean diffusivity and NM-MRI contrast-to-noise ratios (r=-0.44). In both analyses, the strongest effects were observed in the ventral SN-VTA.
Conclusions: These findings suggest that lower levels of non-neuromelanin-bound iron in the SN-VTA contribute to striatal hyperdopaminergia in schizophrenia. Further investigation is warranted to understand the role of low iron in schizophrenia and its potential as a treatment target.
Keywords: Biological Markers; Brain Imaging Techniques; Neuroanatomy; Neurocircuitry; Neuroimaging; Schizophrenia Spectrum and Other Psychotic Disorders.
Figures
References
-
- Trujillo P, Summers PE, Ferrari E, et al. Contrast mechanisms associated with neuromelanin-MRI: Neuromelanin-MRI Contrast. Magn Reson Med. 2017;78:1790–1800. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

