Loss of Hepatic Angiotensinogen Attenuates Diastolic Dysfunction in Heart Failure with Preserved Ejection Fraction
- PMID: 40888605
- PMCID: PMC12631940
- DOI: 10.1002/advs.202507554
Loss of Hepatic Angiotensinogen Attenuates Diastolic Dysfunction in Heart Failure with Preserved Ejection Fraction
Abstract
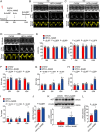
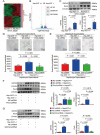
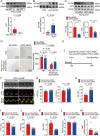
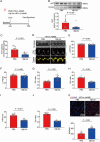
Heart failure with preserved ejection fraction (HFpEF) is a prevalent complex syndrome characterized by diastolic dysfunction with limited therapeutic options. While the renin-angiotensin system (RAS) is implicated in heart failure pathogenesis, the causal contribution of angiotensinogen (AGT), the unique precursor of the RAS, to HFpEF remains undefined. Using a two-hits mouse HFpEF model (high-fat diet + L-NAME), consistent upregulation of hepatic and plasma AGT is identified in wild-type mice of both sexes. Critically, hepatocyte-specific AGT deletion directly ameliorated diastolic dysfunction in male and female HFpEF mice, whereas systemic angiotensin II blockade (losartan) failed to improve cardiac diastolic function. Mechanistically, hepatic AGT drove HFpEF through LRP2-mediated internalization in cardiac endothelial cells, suppressing the GATA2/Pim3 signaling axis, which inhibited microvascular angiogenesis and ultimately exacerbated diastolic dysfunction. To validate therapeutic potential, it is demonstrated that 18β-glycyrrhetinic acid - identified as a potent hepatic AGT inhibitor - significantly improved cardiac diastolic function in HFpEF mice. These findings establish hepatic AGT as a causal contributor to HFpEF pathogenesis and reveal its therapeutic targeting as a promising strategy.
Keywords: 18β‐glycyrrhetinic acid; angiotensin II‐independent mechanism; angiotensinogen; heart failure with preserved ejection fraction; microvascular angiogenesis.
© 2025 The Author(s). Advanced Science published by Wiley‐VCH GmbH.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




References
-
- Tsao C. W., Aday A. W., Almarzooq Z. I., Anderson C. A. M., Arora P., Avery C. L., Baker‐Smith C. M., Beaton A. Z., Boehme A. K., Buxton A. E., Commodore‐Mensah Y., Elkind M. S. V., Evenson K. R., Eze‐Nliam C., Fugar S., Generoso G., Heard D. G., Hiremath S., Ho JE., Kalani R., Kazi D. S., Ko D., Levine D. A., Liu J., Ma J., Magnani J. W., Michos E. D., Mussolino M. E., Navaneethan S. D., Parikh N. I., et al., Circulation 2023, 147, 93.
-
- Bozkurt B., Coats A. J. S., Tsutsui H., Abdelhamid C. M., Adamopoulos S., Albert N., Anker S. D., Atherton J., Böhm M., Butler J., Drazner M. H., Felker G. M., Filippatos G., Fiuzat M., Fonarow G. C., Gomez‐Mesa J. E., Heidenreich P., Imamura T., Jankowska E. A., Januzzi J., Khazanie P., Kinugawa K., Lam CSP, Matsue Y., Metra M., Ohtani T., Francesco M. P., Ponikowski P., Rosano G. M. C., Sakata Y., et al., Eur. J. Heart Fail 2021, 23, 352. - PubMed
-
- McDonagh T. A., Metra M., Adamo M., Gardner R. S., Baumbach A., Böhm M., Burri H., Butler J., Celutkiene J., Chioncel O., Cleland J. G. F., Coats A. J. S., Crespo‐Leiro M. G., Farmakis D., Gilard M., Heymans S., Hoes A. W., Jaarsma T., Jankowska E. A., Lainscak M., Lam C. S. P., Lyon A. R., McMurray J. J. V., Mebazaa A., Mindham R., Muneretto C., Francesco Piepoli M., Price S., Rosano G. M. C., Ruschitzka F., Eur. Heart J. 2021, 42, 3599.
-
- Heidenreich P. A., Bozkurt B., Aguilar D., Allen L. A., Byun J. J., Colvin M. M., Deswal A., Drazner M. H., Dunlay S. M., Evers L. R., Fang J. C., Fedson S. E., Fonarow G. C., Hayek S. S., Hernandez A. F., Khazanie P., Kittleson M. M., Lee C. S., Link M. S., Milano C. A., Nnacheta L. C., Sandhu A. T., Stevenson L. W., Circulation 2022, 145, 876. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous
