This is a preprint.
Profiling a large HIV-1 elite neutralizer cohort reveals remarkable CD4bs bNAb for HIV-1 prevention and therapy
- PMID: 40909752
- PMCID: PMC12407976
- DOI: 10.1101/2025.08.27.672638
Profiling a large HIV-1 elite neutralizer cohort reveals remarkable CD4bs bNAb for HIV-1 prevention and therapy
Update in
-
Profiling of HIV-1 elite neutralizer cohort reveals a CD4bs bnAb for HIV-1 prevention and therapy.Nat Immunol. 2025 Nov;26(11):2016-2029. doi: 10.1038/s41590-025-02286-5. Epub 2025 Oct 6. Nat Immunol. 2025. PMID: 41053396 Free PMC article.
Abstract
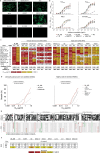
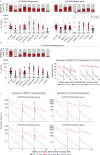
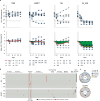
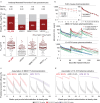
Administration of HIV-1 neutralizing antibodies can suppress viremia and prevent infection in vivo. However, clinical use is challenged by broad envelope sequence diversity and rapid emergence of viral escape1-9. Here, we performed single B cell profiling of 32 top HIV-1 elite neutralizers to identify broadly neutralizing antibodies (bNAbs) with highest potency and breadth for clinical application. From 831 expressed monoclonal antibodies, we identified 04_A06, a new VH1-2-encoded CD4 binding site bNAb with remarkable breadth and potency against extended multiclade pseudovirus panels (GeoMean IC50 = 0.059 μg/ml, breadth = 98.5%, 332 virus strains). Moreover, 04_A06 was not susceptible to classic viral CD4bs escape variants and maintained full viral suppression in HIV-1-infected humanized mice. Structural analyses revealed that antiviral activity is mediated by an unusually long 11-amino acid heavy chain insertion. This insertion facilitates inter-protomer contacts and interactions with highly conserved residues on the adjacent gp120 protomer. Finally, 04_A06 demonstrated high activity against contemporaneously circulating viruses from the Antibody Mediated Prevention (AMP) trials (GeoMean IC50 = 0.082 μg/ml, breadth = 98.4%, 191 virus strains) and in silico modeling for 04_A06LS predicted HIV-1 prevention efficacy of >93%. Thus, 04_A06 will provide unique opportunities for effective treatment and prevention strategies of HIV-1 infection.
Conflict of interest statement
Competing interest declaration A patent application that comprises aspects of this work has been filed by the University of Cologne, listing L.G. and F.K. as inventors. H.G., P.S., and F.K. are listed as inventors on additional patent applications on HIV-1 neutralizing antibodies and have received payments from the University of Cologne for licensed patents. JDB and CER are inventors on Fred Hutch licensed patents related to viral deep mutational scanning. JDB consults for Apriori Bio.
Figures

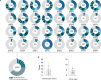










References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
