PAC1 Receptor Knockout Mice Reveal Critical Links Between ER Stress, Myelin Homeostasis, and Neurodegeneration
- PMID: 40943590
- PMCID: PMC12428991
- DOI: 10.3390/ijms26178668
PAC1 Receptor Knockout Mice Reveal Critical Links Between ER Stress, Myelin Homeostasis, and Neurodegeneration
Abstract
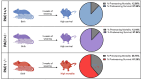
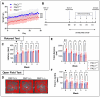
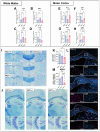
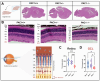
The pituitary adenylate cyclase-activating polypeptide receptor 1 (PAC1) plays a pivotal role in central nervous system development and homeostasis. Comparisons of PAC1 knockout (PAC1-/-), heterozygous (PAC1+/-) and wild-type (PAC1+/+) mice demonstrate that PAC1 deficiency severely impairs pre-weaning survival and results in marked developmental deficits, including reduced postnatal weight and altered locomotor behavior. PAC1-/- mice exhibited hyperlocomotion, reduced anxiety-like behavior, and transient deficits in motor coordination. Gene expression analyses revealed widespread dysregulation of oligodendrocyte-associated markers, with significant myelin reduction and decreased mature oligodendrocyte density in the corpus callosum. ER stress was evidenced in both white matter and motor cortex, as indicated by altered expression of UPR-related genes and increased phosphorylated (p)IRE1+ neurons. Retinal morphology was compromised in PAC1-/- animals, with reduced overall retinal and ganglion cell layer thickness. Notably, no gross morphological or molecular abnormalities were detected in the spinal cord regarding myelin content or MBP expression; however, synaptic marker expression was selectively reduced in the ventral horn of PAC1-deficient mice. Together, these findings highlight a critical role for PAC1 in oligodendrocyte maturation, retinal development, and synaptogenesis, providing new insights with relevance in multiple sclerosis and other neurodevelopmental and neurodegenerative conditions.
Keywords: PAC1 receptor; endoplasmic reticulum (ER) stress 2; multiple sclerosis; myelin homeostasis; neurodegeneration; oligodendrocytes; unfolded protein response (UPR).
Conflict of interest statement
The authors declare no conflicts of interest.
Figures


References
-
- Walton C., King R., Rechtman L., Kaye W., Leray E., Marrie R.A., Robertson N., La Rocca N., Uitdehaag B., van der Mei I., et al. Rising prevalence of multiple sclerosis worldwide: Insights from the Atlas of MS, third edition. Mult. Scler. J. 2020;26:1816–1821. doi: 10.1177/1352458520970841. - DOI - PMC - PubMed
-
- Elliott C., Belachew S.M., Wolinsky J.S., Hauser S.L., Kappos L., Barkhof F., Bernasconi C., Fecker J., Model F., Wei W., et al. Chronic white matter lesion activity predicts clinical progression in primary progressive multiple sclerosis. Brain. 2019;142:2787–2799. doi: 10.1093/brain/awz212. - DOI - PMC - PubMed
-
- Dhaiban S., Al-Ani M., Elemam N.M., Al-Aawad M.H., Al-Rawi Z., Maghazachi A.A. Role of Peripheral Immune Cells in Multiple Sclerosis and Experimental Autoimmune Encephalomyelitis. Sci. 2021;3:12. doi: 10.3390/sci3010012. - DOI
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

