This is a preprint.
Translational reading frame determines the pathogenicity of C-terminal frameshift deletions in MeCP2: an alternative therapeutic approach
- PMID: 40949944
- PMCID: PMC12424628
- DOI: 10.1101/2025.08.31.673354
Translational reading frame determines the pathogenicity of C-terminal frameshift deletions in MeCP2: an alternative therapeutic approach
Abstract
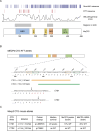
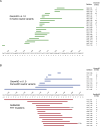
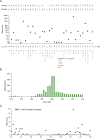
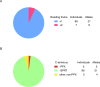
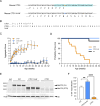
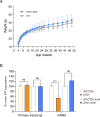
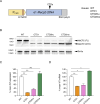
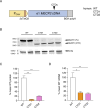
Mutations in the MECP2 gene cause the severe neurological disorder Rett syndrome. A cluster of frameshift-causing C-terminal deletions (CTDs) lead to loss of ~100 amino acids at the C-terminus of the MeCP2 protein, and account for approximately 10% of RTT-causing mutations. The pathogenicity of C-terminal deletions (CTDs) is unexpected, as this C-terminal domain is non-essential in mice. Utilising databases of pathogenic and benign human MECP2 mutations, we find that some individuals with apparently typical CTDs do not exhibit Rett syndrome, confirming that C-terminal truncations are not intrinsically pathogenic. Using human DNA sequence data and mouse models, we demonstrate that pathogenicity results from a drastic reduction in MeCP2 levels and is determined by the presence of the short amino acid motif proline-proline-stop (-PPX) at the C-terminus, which results from a shift to the +2 reading frame. Individuals with CTDs that shift to the +1 frame avoid this motif and do not develop Rett syndrome. Mutating the stop codon of the PPX motif to tryptophan rescues MeCP2 expression and RTT-like phenotypes in a CTD mouse model. Finally, we demonstrate that an adenine base editor can efficiently introduce this tryptophan substitution in cultured cells. Overall, our findings uncover a simple and reliable prognostic distinction between benign and pathogenic CTDs and provide proof-of-concept for an editing strategy that potentially corrects all disease-causing CTD mutations.
Conflict of interest statement
Competing Interests Statement B.P.K. is an inventor on patents or patent applications filed by Mass General Brigham that describe genome engineering technologies, including the SpG and SpRY enzymes. B.P.K. has consulted for EcoR1 capital, Novartis Venture Fund, Foresite Labs, and Jumble Therapeutics, and is on the scientific advisory boards of Acrigen Biosciences, Life Edit Therapeutics, and Prime Medicine. B.P.K. has a financial interest in Prime Medicine, Inc., a company developing therapeutic CRISPR-Cas technologies for gene editing. B.P.K.’s interests were reviewed and are managed by MGH and MGB in accordance with their conflict-of-interest policies.
Figures





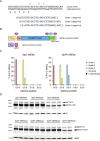
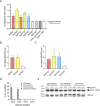

References
-
- Alves CRR, Ha LL, Yaworski R, Sutton ER, Lazzarotto CR, Christie KA, Reilly A, Beauvais A, Doll RM, de la Cruz D, Maguire CA, Swoboda KJ, Tsai SQ, Kothary R, Kleinstiver BP. 2024. Optimization of base editors for the functional correction of SMN2 as a treatment for spinal muscular atrophy. Nat Biomed Eng 8:118–131. doi: 10.1038/s41551-023-01132-z - DOI - PMC - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
