Disrupted cholesterol biosynthesis and hair follicle stem cell impairment in the onset of alopecia
- PMID: 40953083
- PMCID: PMC12435693
- DOI: 10.1371/journal.pone.0308455
Disrupted cholesterol biosynthesis and hair follicle stem cell impairment in the onset of alopecia
Abstract
Background: Hair follicle cycle and the functioning of stem cells in alopecia are influenced by the suppression of cholesterol synthesis and the accumulation of sterol intermediates.
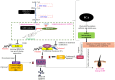
Objective: This investigation aims to elucidate the regulatory function of disrupted cholesterol homeostasis in the functioning of hair follicle stem cells (HFSCs) and the cycling of hair follicles. Additionally, it seeks to provide an understanding of the fate of stem cells in primary cicatricial alopecia (PCA).
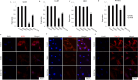
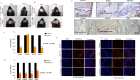
Methods: To evaluate the influence of cholesterol on the functionality of hair follicles, a study was conducted to analyse gene expression and pathways associated with hair follicle stem cell markers in scalp samples affected by PCA (LPP, FFA, CCCA, DC, DF, TF). To assess the influence of disrupted cholesterol homeostasis on HFSCs, we conducted experiments involving the administration of 7-dehydrocholesterol (7DHC) and BM15766 (Pharmacological inhibitor of cholesterol biosynthesis), to Human Hair Follicle Outer Root Sheath Cells (HORSCs), as well as C57BL/6 mice, and hair follicle organoid cultures. The study utilised reverse transcription polymerase chain reaction (RT-PCR) to assess gene expression, while immunofluorescence was employed to analyse protein expression. The tracking of stem cell fate was accomplished through the utilisation of a BrdU pulse-chase experiment, while the verification of apoptotic consequences was established by utilising the TUNEL assay. A statistical analysis was conducted to assess the statistical significance of the data.
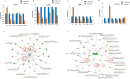
Results: There was a notable decrease in the expression of HFSC marker genes among patients afflicted with PCA. In vitro data further confirmed the cholesterol inhibition and sterol intermediate accumulation in stem cells, leading to stem cell characteristics' disruption. The experimental group of mice exposed to 7DHC and BM15766 also exhibited a notable inability to initiate hair regrowth. Consequently, this deficiency in hair regrowth resulted in the activation of apoptosis, specifically in the stem cells. Additionally, our confirmatory analysis, which was performed utilising organoid culture, consistently yielded comparable results. The results as mentioned above emphasise the significant importance of cholesterol production in preserving the integrity and functionality of HFSCs, hence providing novel insights into the progression of alopecia.
Conclusion: Individuals with aberrant cholesterol production, especially those affected by PCA, endure a lasting hindrance in hair regrowth stemming from the irreversible damage to their hair follicles. The observed phenomenon is hypothesized to result from the depletion of hair follicle stem cells. Our study reveals further insights that unveil the previously unrecognized role of sterol intermediates in governing hair follicle cycling and stem cell function in PCA. The modulation of cholesterol production and the accumulation of sterol intermediates significantly influence the hair follicle cycle and the role of stem cells in alopecia.
Copyright: © 2025 Nikhila et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures

References
-
- Roh C, Lyle S. Cutaneous stem cells and wound healing. Pediatr Res. 2006;59(4 Pt 2):100R–3R. Epub 2006/03/22. doi: 10.1203/01.pdr.0000203572.51876.ba - DOI - PubMed
-
- Panicker SP, Ganguly T, Consolo M, Price V, Mirmirani P, Honda K, et al. Sterol intermediates of cholesterol biosynthesis inhibit hair growth and trigger an innate immune response in cicatricial alopecia. PLoS One. 2012;7(6):e38449. Epub 2012/06/12. doi: 10.1371/journal.pone.0038449 ; PMCID: PMCPMC3369908 - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

