Cell type-specific plasticity in synaptic, intrinsic, and sound response properties of deep-layer cortical neurons after noise trauma
- PMID: 40971419
- PMCID: PMC12448056
- DOI: 10.1126/sciadv.adx9737
Cell type-specific plasticity in synaptic, intrinsic, and sound response properties of deep-layer cortical neurons after noise trauma
Abstract
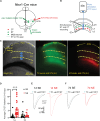
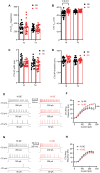
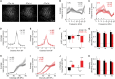
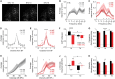
Peripheral damage drives auditory cortex (ACtx) plasticity, but the underlying synaptic and cellular mechanisms remain poorly understood. We used a combination of in vitro slice electrophysiology, optogenetics, and in vivo two-photon imaging to investigate layer 5 extratelencephalic (ET) and layer 6 corticothalamic (CT) neuronal plasticity in mice, following noise-induced hearing loss (NIHL). Thalamocortical input was initially balanced between CTs and ETs but shifted to CT-dominant 1 day post-NIHL and then normalized by day 7. This transient shift was accompanied by increased quantal size and suprathreshold excitability in CTs, with minimal changes in ETs. In vivo, CTs exhibited persistent elevation in sound intensity thresholds, while ETs showed a transient shift in frequency tuning and reduced high-frequency responsiveness that recovered within a week. These findings reveal distinct, cell type-specific plasticity mechanisms in deep-layer ACtx neurons following peripheral damage and highlight potential targets for treating hearing loss-related disorders such as tinnitus and hyperacusis.
Figures



Update of
-
Cell-type-specific plasticity in synaptic, intrinsic, and sound response properties of deep-layer auditory cortical neurons after noise trauma.bioRxiv [Preprint]. 2025 Aug 15:2025.07.09.663954. doi: 10.1101/2025.07.09.663954. bioRxiv. 2025. Update in: Sci Adv. 2025 Sep 19;11(38):eadx9737. doi: 10.1126/sciadv.adx9737. PMID: 40791381 Free PMC article. Updated. Preprint.
References
-
- Rasmusson D. D., Reorganization of raccoon somatosensory cortex following removal of the fifth digit. J. Comp. Neurol. 205, 313–326 (1982). - PubMed
-
- Merzenich M. M., Kaas J. H., Wall J., Nelson R. J., Sur M., Felleman D., Topographic reorganization of somatosensory cortical areas 3b and 1 in adult monkeys following restricted deafferentation. Neuroscience 8, 33–55 (1983). - PubMed
-
- Gilbert C. D., Wiesel T. N., Receptive field dynamics in adult primary visual cortex. Nature 356, 150–152 (1992). - PubMed
-
- Kaas J. H., Krubitzer L. A., Chino Y. M., Langston A. L., Polley E. H., Blair N., Reorganization of retinotopic cortical maps in adult mammals after lesions of the retina. Science 248, 229–231 (1990). - PubMed
-
- Robertson D., Irvine D. R., Plasticity of frequency organization in auditory cortex of guinea pigs with partial unilateral deafness. J. Comp. Neurol. 282, 456–471 (1989). - PubMed
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials

