Ex vivo expansion of corneal endothelial cells enabled by small molecule inhibitors of LATS kinase
- PMID: 40979555
- PMCID: PMC12445230
- DOI: 10.1016/j.reth.2025.08.014
Ex vivo expansion of corneal endothelial cells enabled by small molecule inhibitors of LATS kinase
Abstract
Introduction: Transplantation of expanded corneal endothelial cells (CECs) has been regarded as a promising approach for treating corneal diseases caused by CEC damage or dysfunction. However, an efficient method for expanding CECs remains inadequately established.
Methods: We examined whether small molecule inhibitors of large tumor suppressor kinase (LATS) promote the proliferation of CECs. We also evaluated the expression of functional markers in CECs treated with the inhibitors.
Results: We found that LATS kinase inhibitors enhance the cell density of bovine CECs ex vivo. CECs that were expanded in the presence of these inhibitors exhibited increased nuclear translocation of yes-associated protein (YAP) and upregulated expression of YAP-regulated genes. Furthermore, we observed that YAP was essential for promoting cell proliferation. Notably, the inhibitors also increased the density of primary human CECs. Expanded human CECs expressed CEC functional markers, including Na+/K+-transporting ATPase subunit alpha-1 (ATP1A1), Zonula occludens-1 (ZO-1), and N-cadherin; they showed upregulated expression of YAP-regulated genes.
Conclusions: Collectively, these findings support the development of efficient culture techniques for CEC expansion and may facilitate the advancement of therapeutic strategies for CEC-associated diseases.
Keywords: Corneal endothelial cells; Ex vivo expansion; Hippo pathway; LATS inhibitors.
© 2025 The Author(s).
Conflict of interest statement
The authors declare the following financial interests/personal relationships which may be considered as potential competing interests:Ryuhei Hayashi reports financial support was provided by the 10.13039/100009619Japan Agency for Medical Research and Development (10.13039/100009619AMED), the 10.13039/501100002241Japan Science and Technology Agency, and the 10.13039/501100001691Japan Society for the Promotion of Science (JSPS). If there are other authors, they declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures




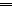 N). (E) BCE C/D-1b cells were cultured in low-attachment U-bottom plates with GA-002, GA-017, and/or Verteporfin (YAP-TEAD interaction inhibitor) for 4 days. Cell numbers were assessed using an ATP assay and normalized to day 0. The y-axis represents the relative cell number (% of DMSO control without Verteporfin). (F) Relative luciferase activity in BCE C/D-1b cells transfected with a pGL4.20-GTIIC YAP-responsive reporter. Cells were treated with DMSO, 10 μM GA-002 or GA-017 for 1 day in the presence or absence of 1 μM Verteporfin. Signals were normalized to cell number assessed by WST assay. The y-axis indicates relative luciferase activity compared with DMSO control without Verteporfin. Data represent means ± SD of three independent experiments. Statistical significance was analyzed using Dunnett's test for (A), (B), and (D), or Tukey's test for (C) and (E). ∗P < 0.05, ∗∗P < 0.01, ∗∗∗P < 0.001. N.S., not significant.
N). (E) BCE C/D-1b cells were cultured in low-attachment U-bottom plates with GA-002, GA-017, and/or Verteporfin (YAP-TEAD interaction inhibitor) for 4 days. Cell numbers were assessed using an ATP assay and normalized to day 0. The y-axis represents the relative cell number (% of DMSO control without Verteporfin). (F) Relative luciferase activity in BCE C/D-1b cells transfected with a pGL4.20-GTIIC YAP-responsive reporter. Cells were treated with DMSO, 10 μM GA-002 or GA-017 for 1 day in the presence or absence of 1 μM Verteporfin. Signals were normalized to cell number assessed by WST assay. The y-axis indicates relative luciferase activity compared with DMSO control without Verteporfin. Data represent means ± SD of three independent experiments. Statistical significance was analyzed using Dunnett's test for (A), (B), and (D), or Tukey's test for (C) and (E). ∗P < 0.05, ∗∗P < 0.01, ∗∗∗P < 0.001. N.S., not significant.

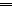 N). (E) Levels of YAP phosphorylation in human primary CECs treated with DMSO, GA-002, or GA-017 for 1 h, assessed using a HTRF assay. NT: non-treatment; DMSO: vehicle control; GA-002 or GA-017: 10 μM. Data represent means ± SD from 4 to 6 donors. Statistical significance was assessed using Tukey's test for (B) and (D), or Dunnett's test for (E). ∗P < 0.05, ∗∗P < 0.01, ∗∗∗P < 0.001.
N). (E) Levels of YAP phosphorylation in human primary CECs treated with DMSO, GA-002, or GA-017 for 1 h, assessed using a HTRF assay. NT: non-treatment; DMSO: vehicle control; GA-002 or GA-017: 10 μM. Data represent means ± SD from 4 to 6 donors. Statistical significance was assessed using Tukey's test for (B) and (D), or Dunnett's test for (E). ∗P < 0.05, ∗∗P < 0.01, ∗∗∗P < 0.001.References
-
- Joyce N.C., Meklir B., Joyce S.J., Zieske J.D. Cell cycle protein expression and proliferative status in human corneal cells. Investig Ophthalmol Vis Sci. 1996;37:645–655. - PubMed
LinkOut - more resources
Full Text Sources
Research Materials

