Vorapaxar enhanced mitochondria-associated ferroptosis primes cancer immunotherapy via targeting FOXO1/HMOX1 axis
- PMID: 41005298
- PMCID: PMC12629800
- DOI: 10.1016/j.xcrm.2025.102371
Vorapaxar enhanced mitochondria-associated ferroptosis primes cancer immunotherapy via targeting FOXO1/HMOX1 axis
Abstract
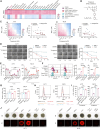
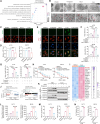
Immunotherapy has revolutionized cancer treatment, yet challenges persist, such as resistance and lethal thromboembolism, necessitating dual-purpose strategies. Targeting ferroptosis emerges as a promising strategy to enhance immunotherapy efficacy, prompting our investigation of antiplatelet agents that simultaneously promote ferroptosis and mitigate thromboembolic risks. Through systematic screening of 20 Food and Drug Administration (FDA)-approved antiplatelet agents, we identify vorapaxar as a potent pro-ferroptotic drug. Mechanistically, vorapaxar binds forkhead box O1 (FOXO1), inhibits its phosphorylation at Ser256, and facilitates nuclear translocation to upregulate heme oxygenase 1 (HMOX1), promoting mitochondrial iron overload and mitochondria-associated ferroptosis. Vorapaxar enhances immunotherapy-induced tumor ferroptosis and antitumor immunity across diverse melanoma models, including B16F10 tumor-bearing mice, Braf/Pten-driven spontaneous melanoma mice, and peripheral blood mononuclear cell (PBMC)-humanized mice. Clinically, high FOXO1/HMOX1 co-expression correlates with improved immunotherapy response and progression-free survival. These findings position vorapaxar as a promising adjunct to immunotherapy, offering a dual benefit for cancer patients requiring both antithrombotic therapy and immunotherapy.
Keywords: FOXO1; HMOX1; ferroptosis; immunotherapy; vorapaxar.
Copyright © 2025 The Authors. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of interests The authors declare no competing interests.
Figures





References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

