Exosome-like Nanoparticles Extracted from Plant Cells for Diabetes Therapy
- PMID: 41009713
- PMCID: PMC12470572
- DOI: 10.3390/ijms26189155
Exosome-like Nanoparticles Extracted from Plant Cells for Diabetes Therapy
Abstract
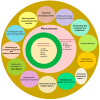
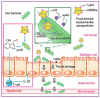
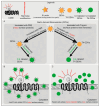
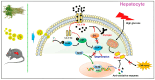
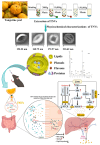
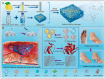
Diabetes mellitus (DM) is a complex metabolic disorder characterized by chronic hyperglycemia and associated complications such as cardiovascular disease, nephropathy, retinopathy, neuropathy, and chronic non-healing wounds. Current antidiabetic therapies offer only partial glycemic control and are limited by poor bioavailability, adverse effects, and an inability to prevent disease progression. Plant-derived exosome-like nanoparticles (PENPs) have emerged as a promising class of natural nanocarriers with excellent biocompatibility, low immunogenicity, and intrinsic multi-component bioactivity. However, few reviews have addressed recent progress in PENPs for DM therapy. To capture the recent developments in this area, this review provides a systematic synthesis of recent advances in PENPs for DM therapy, covering plant sources, extraction and purification methods, molecular compositions, and therapeutic mechanisms. Preclinical studies have demonstrated that PENPs can improve hyperglycemia, enhance insulin sensitivity, regulate hepatic lipid metabolism, and promote wound healing by modulating oxidative stress, inflammation, gut microbiota, glucose metabolism, and insulin signaling. Additionally, PENPs have been shown to promote angiogenesis via glycolytic reprogramming. Despite these promising findings, challenges including scalable isolation, standardized physicochemical characterization, and clinical translation remain. Future directions include engineering multifunctional PENPs, establishing Good Manufacturing Practice (GMP)-compliant production, and conducting clinical trials to facilitate their integration into precision therapeutics for diabetes management.
Keywords: diabetes and its complications; nanobiomedicine; natural nanocarriers; plant-derived exosome-like nanoparticles; precision therapy.
Conflict of interest statement
The authors report no conflicts of interest.
Figures





References
-
- Ong K.L., Stafford L.K., McLaughlin S.A., Boyko E.J., Vollset S.E., Smith A.E., Dalton B.E., Duprey J., Cruz J.A., Hagins H., et al. GBD 2021 Diabetes Collaborators: Global, regional, and national burden of diabetes from 1990 to 2021, with projections of prevalence to 2050: A systematic analysis for the Global Burden of Disease Study 2021. Lancet. 2023;402:203–234. doi: 10.1016/S0140-6736(23)01301-6. Erratum in Lancet 2025, 405, 202. - DOI - PMC - PubMed
-
- International Diabetes Federation . IDF Diabetes Atlas. 11th ed. International Diabetes Federation; Brussels, Belgium: 2025. [(accessed on 4 July 2025)]. Available online: https://idf.org/news/idf-diabetes-atlas-11th-edition.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

