Mitigating Pro-Inflammatory SASP and DAMP With Urolithin A: A Novel Senomorphic Strategy
- PMID: 41021772
- PMCID: PMC12608089
- DOI: 10.1111/acel.70237
Mitigating Pro-Inflammatory SASP and DAMP With Urolithin A: A Novel Senomorphic Strategy
Abstract
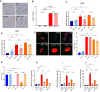
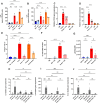
Senescent cells are known to contribute to aging and age-related diseases. One key way they influence aging is by secreting senescence-associated secretory phenotype (SASP) factors along with several damage-associated molecular pattern (DAMP) molecules. Consequently, inhibiting SASP and DAMP signaling (senomorphics) has emerged as a therapeutic strategy. Urolithin A (UA), a gut-derived metabolite produced from ellagitannins and ellagic acid found in berries, nuts, and pomegranates, has demonstrated potent anti-inflammatory properties and protective effects against aging and age-related conditions in experimental models. Here we demonstrate that UA lowers the expression and release of pro-inflammatory SASP and DAMP factors, at least in part, by downregulating cytosolic DNA release and subsequent decrease in cGAS-STING signaling.
Keywords: Urolithin A; cGAS‐STING signaling; cytosolic DNA; senescence; senomorphic.
© 2025 The Author(s). Aging Cell published by Anatomical Society and John Wiley & Sons Ltd.
Conflict of interest statement
M.C. and J.K.A. are co‐founders of Symbiont Bio and declares no conflicts of interest.
Figures
References
-
- Admasu, T. D. , Kim K., Rae M., et al. 2023. “Selective Ablation of Primary and Paracrine Senescent Cells by Targeting Iron Dyshomeostasis.” Cell Reports 42: 112058. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

