Synthesis-Driven Functionality in High-Entropy Materials
- PMID: 41026746
- PMCID: PMC12590539
- DOI: 10.1002/smll.202501703
Synthesis-Driven Functionality in High-Entropy Materials
Abstract
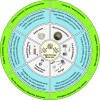
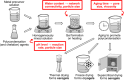
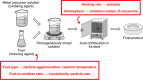
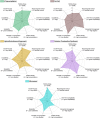
Since their discovery in 2015, high-entropy oxides have introduced a paradigm shift in materials science, unveiling a class of compounds with exceptional structural and functional versatility. These high-entropy materials (HEMs) offer exciting opportunities as next-generation alternatives to conventional materials, owing to the synergistic interplay of multiple principal elements that results in enhanced stability, tunability, and multifunctionality. Their unique atomic configurations enable the design of materials with high surface areas and abundant active sites for catalysis, mechanically robust structures for energy storage, or tunable band gaps for electronic and optoelectronic devices. However, the vast compositional space of HEMs presents both a challenge and an opportunity. Meaningful property design requires a deep understanding of how synthesis routes influence structure-property relationships. In this review, a comprehensive overview of established and emerging synthesis strategies for HEMs, focusing on how each method affects resulting structural, electronic, electrochemical, and optical characteristics, is provided. Key process parameters that can be tailored to optimize material performance are highlighted. Additionally, the accelerating role of high-throughput synthesis and characterization in navigating the design space of high-entropy systems is discussed. By systematically connecting synthesis, structure, and function, this review aims to guide the rational design of HEMs for energy applications and beyond.
Keywords: electronic applications; energy applications; high‐entropy materials; high‐entropy oxides; synthesis techniques.
© 2025 The Author(s). Small published by Wiley‐VCH GmbH.
Conflict of interest statement
The authors declare no conflict of interest.
Figures







References
-
- Cantor B., Chang I. T. H., Knight P., Vincent A. J. B., Mater. Sci. Eng. A 2004, 375–377, 213.
-
- Yeh J.‐W., Chen S.‐K., Lin S.‐J., Gan J.‐Y., Chin T.‐S., Shun T.‐T., Tsau C.‐H., Chang S.‐Y., Adv. Eng. Mater. 2004, 6, 299.
-
- Aamlid S. S., Oudah M., Rottler J., Hallas A. M., J. Am. Chem. Soc. 2023, 145, 5991. - PubMed
-
- Brahlek M., Gazda M., Keppens V., Mazza A. R., McCormack S. J., Mielewczyk‐Gryń A., Musico B., Page K., Rost C. M., Sinnott S. B., Toher C., Ward T. Z., Yamamoto A., APL Mater. 2022, 10, 110902.
-
- Oses C., Toher C., Curtarolo S., Nat. Rev. Mater. 2020, 5, 295.
Publication types
Grants and funding
- 03XP0447/Federal Ministry of Education and Research
- HA1344-45/German Research Foundation
- 101017709/European Union's Horizon 2020 research and innovation program
- "3DMatterMadetoOrder"(EXC-2082/1-390761711)/DFG under Germany's Excellence Strategy via the Excellence Cluster
- Auto.MAP/Helmholtz-Gemeinschaft
LinkOut - more resources
Full Text Sources

