Prognostic value of systemic inflammation markers in early stage non-small cell lung cancer
- PMID: 41028061
- PMCID: PMC12484583
- DOI: 10.1038/s41598-025-08683-y
Prognostic value of systemic inflammation markers in early stage non-small cell lung cancer
Abstract
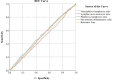
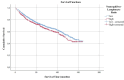
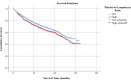
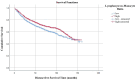
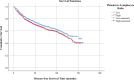
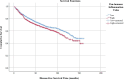
The present study investigates the prognostic significance of systemic inflammation markers in patients with early-stage non-small cell lung cancer (NSCLC), who underwent surgical treatment. We retrospectively analyzed data from 2,159 patients, who underwent lung resection for stage I-IIA NSCLC in nine centers between the years 2010 and 2022. In order to compare survival, we grouped the patients by preoperative neutrophil-to-lymphocyte ratio (NLR), lymphocyte-to-monocyte ratio (LMR), platelet-to-lymphocyte ratio (PLR), and pan-immune inflammation value (PIV). The mean overall survival (OS) was significantly shorter in patients with high NLR (102.7 vs. 109.4 months, p = 0.040). A low LMR was associated with worse OS (101 vs. 110.3 months, p < 0.001) and worse disease-free survival (DFS) (100.2 vs. 108.6 months, p = 0.020). A high PLR was a poor prognostic factor for both OS (104.1 vs. 110.1 months, p = 0.017) and DFS (102.5 vs. 108.7 months, p = 0.021). A high PIV was associated with worse DFS (101.2 vs. 109.8 months, p = 0.003). Patients with high PIV had longer time to chest tube removal (6.9 vs. 6.7 days, p = 0.049) and longer hospital stays (8.6 vs. 8.2 days, p < 0.001). Complication rates were higher in patients with lower LMR (33.8% vs. 29.4%, p = 0.028) and higher PLR (38.1% vs. 33.1%, p = 0.016). In our multicenter study, OS was worse in patients with high PLR, high NLR, and low LMR. DFS was worse in patients with high PIV, high PLR, and low LMR. NLR, LMR, PLR, and PIV were associated with OS and DFS in early-stage NSCLC, but none of the circulating inflammatory markers proved to be statistically significant in the multivariate analysis.
Keywords: Early-stage NSCLC; Lung cancer; Prognosis; Systemic inflammation.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Competing interests: The authors declare no competing interests. Human ethics and consent to participate declarations: We obtained written informed consent from each patient and conducted the study in accordance with the principles of the Declaration of Helsinki. Ege University Medical Research Ethics Committee granted approval for the study (No: 23-12T/20).
Figures



References
-
- Sung, H. et al. Global Cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin.71 (3), 209–249 (2021). - PubMed
-
- Donnem, T. et al. Strategies for clinical implementation of TNM-Immunoscore in resected nonsmall-cell lung cancer. Ann. Oncol.27 (2), 226–232 (2016). - PubMed
-
- Shin, J. et al. Prognostic impact of newly proposed M descriptors in TNM classification of Non-Small cell lung Cancer. J. Thorac. Oncol.12 (3), 520–528 (2017). - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

