Emerging roles for complement in lung transplantation
- PMID: 41031895
- PMCID: PMC12483610
- DOI: 10.1172/JCI188346
Emerging roles for complement in lung transplantation
Abstract
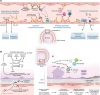
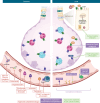
The complement system is an evolutionarily conserved host defense system that has evolved from invertebrates to mammals. Over time, this system has become increasingly appreciated as having effects beyond purely bacterial clearance, with clinically relevant implications in transplantation, particularly lung transplantation. For many years, complement activation in lung transplantation was largely focused on antibody-mediated injuries. However, recent studies have highlighted the importance of both canonical and noncanonical complement activation in shaping adaptive immune responses, which influence alloimmunity. These studies, together with the emergence of FDA-approved complement therapeutics and other drugs in the pipeline that function at different points of the cascade, have led to an increased interest in regulating the complement system to improve donor organ availability as well as improving both short- and long-term outcomes after lung transplantation. In this Review, we provide an overview of the when, what, and how of complement in lung transplantation, posing the questions of: when does complement activation occur, what components of the complement system are activated, and how can this activation be controlled? We conclude that complement activation occurs at multiple stages of the transplant process and that randomized controlled trials will be necessary to realize the therapeutic potential of neutralizing this activation to improve outcomes after lung transplantation.
Conflict of interest statement
Figures

References
-
- Singh TP, et al. The international thoracic organ transplant registry of the International Society for Heart and Lung Transplantation: 2025 Annual Report of Heart and Lung Transplantation. J Heart Lung Transplant. In press. - PubMed
-
- Singh TP, et al. Graft survival in primary thoracic organ transplant recipients: A special report from the International Thoracic Organ Transplant Registry of the International Society for Heart and Lung Transplantation. J Heart Lung Transplant. 2023;42(10):1321–1333. doi: 10.1016/j.healun.2023.07.017. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

