Microglial glycolytic reprogramming in alzheimer's disease: association with impaired phagocytic function and altered vascular proximity
- PMID: 41039597
- PMCID: PMC12492836
- DOI: 10.1186/s12974-025-03546-9
Microglial glycolytic reprogramming in alzheimer's disease: association with impaired phagocytic function and altered vascular proximity
Abstract
Background: Alzheimer's disease (AD) is characterized by chronic neuroinflammation alongside amyloid-beta plaque and phosphorylated tau (p-Tau) tangle accumulation. Microglia, as resident immune cells, undergo glycolytic reprogramming that may exacerbate inflammation and impede toxic protein clearance. Specifically, the glycolytic enzyme pyruvate kinase M2 (PKM2) drives proinflammatory microglial phenotypes linked to neurodegeneration. This study investigates how PKM2-mediated microglial glycolytic reprogramming and inflammatory responses alongside Aβ/p-Tau clearance in human AD brains.
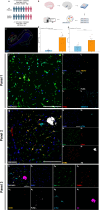
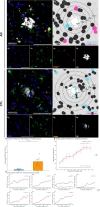
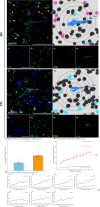
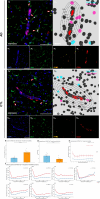
Methods and results: Hippocampal-entorhinal cortex (HP-EC) tissues from 8 AD patients and 8 matched controls underwent multiplex immunohistochemistry and high-resolution spatial analysis. PKM2+Iba1+ microglia density significantly increased in AD versus controls (p < 0.001), predominantly displaying a disease-associated microglial (HAM-like) phenotype (ABCA7+) with concurrent lipid-droplet accumulation (PLIN3+; LDAM phenotype). Spatially, glycolytic PKM2+Iba1+ microglia accumulated near Aβ plaques, p-Tau tangles, and cerebral vasculature. Notably, their distribution around plaques/tau showed anomalous increasing density with distance (p < 0.001), suggesting impaired chemotaxis. Perivascular localization lacked clear chemotactic gradients. Functionally, overall phagocytic activity (CD68+) decreased significantly in AD (p = 0.001), primarily attributed to PKM2- subsets, whereas PKM2+Iba1+ microglia exhibited pronounced phagocytic exhaustion (PLIN2+; p < 0.001), consistent around both Aβ and p-Tau lesions (all p < 0.001).
Conclusion: Our study establishes that microglial glycolytic reprogramming via PKM2 promotes a proinflammatory HAM-like phenotype, phagocytic exhaustion, and peri-pathological accumulation with aggregates and cerebral vessels. Targeting glycolytic pathways represents a viable therapeutic strategy for alleviating microglial dysfunction and neuroinflammation in AD.
Keywords: Alzheimer’s disease; Glycolysis; Microglia; Neuroinflammation; PKM2.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: The research protocol was approved by the Institutional Review Board of the Institute of Basic Medical Sciences, Chinese Academy of Medical Sciences, China (Approval Number: 2022125). Consent for publication: All authors have consented for the publication of manuscript. Competing interests: The authors declare no competing interests.
Figures


References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

