Brain tumors induce widespread disruption of calvarial bone and alteration of skull marrow immune landscape
- PMID: 41044343
- PMCID: PMC12586139
- DOI: 10.1038/s41593-025-02064-4
Brain tumors induce widespread disruption of calvarial bone and alteration of skull marrow immune landscape
Erratum in
-
Author Correction: Brain tumors induce widespread disruption of calvarial bone and alteration of skull marrow immune landscape.Nat Neurosci. 2025 Nov 13. doi: 10.1038/s41593-025-02170-3. Online ahead of print. Nat Neurosci. 2025. PMID: 41233590 No abstract available.
Abstract
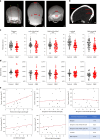
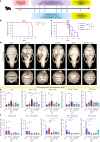
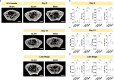
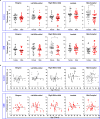
The skull marrow niche has recently been identified as a reservoir that supplies the brain with monocytes and neutrophils in the context of disease and injury, but its role in brain cancers remains unknown. Here we show that glioblastoma, the most malignant type of brain tumor, induces calvarial bone abnormalities in murine models and patients with glioblastoma, altering osteoclast activities and increasing the number of skull channels in mice. Single-cell RNA sequencing revealed glioblastoma-mediated alterations in the immune landscape of skull marrow and femoral bone marrow, including expansion of neutrophils and deterioration of various B cell subsets. In vivo inhibition of bone resorption reduced bone abnormalities, but promoted tumor progression in mesenchymal subtype tumors. This also abolished the survival benefit of the checkpoint inhibitor anti-PD-L1, by reducing activated T cell and increasing inflammatory neutrophil numbers. Together, these data provide insight into how brain tumors affect skull bone and the immune environment.
© 2025. The Author(s).
Conflict of interest statement
Competing interests: The authors declare no competing interests.
Figures












References
MeSH terms
Grants and funding
- S10 OD032169/OD/NIH HHS/United States
- K12 NS080223/NS/NINDS NIH HHS/United States
- P30 CA013330/CA/NCI NIH HHS/United States
- S10 RR029545/RR/NCRR NIH HHS/United States
- 844948/EC | EU Framework Programme for Research and Innovation H2020 | H2020 Priority Excellent Science | H2020 Marie Skłodowska-Curie Actions (H2020 Excellent Science - Marie Skłodowska-Curie Actions)
LinkOut - more resources
Full Text Sources
Medical
Research Materials

