Muscle Cathepsin B Treatment Improves Behavioral and Neurogenic Deficits in a Mouse Model of Alzheimer's Disease
- PMID: 41047763
- PMCID: PMC12610946
- DOI: 10.1111/acel.70242
Muscle Cathepsin B Treatment Improves Behavioral and Neurogenic Deficits in a Mouse Model of Alzheimer's Disease
Abstract
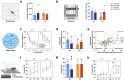
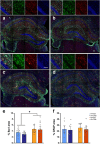
Increasing evidence indicates skeletal muscle function is associated with cognition. Muscle-secreted protease Cathepsin B (Ctsb) is linked to memory in animals and humans, but has an unclear role in neurodegenerative diseases. To address this question, we utilized an AAV-vector-mediated approach to express Ctsb in skeletal muscle of APP/PS1 Alzheimer's disease (AD) model mice. Mice were treated with Ctsb at 4 months of age, followed by behavioral analyses 6 months thereafter. Here we show that muscle-targeted Ctsb treatment results in long-term improvements in motor coordination, memory function, and adult hippocampal neurogenesis, while plaque pathology and neuroinflammation remain unchanged. Additionally, in AD mice, Ctsb treatment normalizes hippocampal, muscle, and plasma proteomic profiles to resemble that of wildtype (WT) controls. In AD mice, Ctsb increases the abundance of hippocampal proteins involved in mRNA metabolism and protein synthesis, including those relevant to adult neurogenesis and memory function. Furthermore, Ctsb treatment enhances plasma metabolic and mitochondrial processes. In muscle, Ctsb treatment elevates protein translation in AD mice, whereas in WT mice mitochondrial proteins decrease. In WT mice, Ctsb treatment causes memory deficits and results in protein profiles across tissues that are comparable to AD control mice. Overall, the biological changes in the treatment groups are consistent with effects on memory function. Thus, skeletal muscle Ctsb application has potential as an AD therapeutic intervention.
Keywords: Alzheimer's disease; cathepsin B; memory; muscle; neurogenesis; proteomics.
© 2025 The Author(s). Aging Cell published by Anatomical Society and John Wiley & Sons Ltd.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures




Update of
-
Muscle Cathepsin B treatment improves behavioral and neurogenic deficits in a mouse model of Alzheimer's Disease.bioRxiv [Preprint]. 2025 Jan 22:2025.01.20.633414. doi: 10.1101/2025.01.20.633414. bioRxiv. 2025. Update in: Aging Cell. 2025 Nov;24(11):e70242. doi: 10.1111/acel.70242. PMID: 39896474 Free PMC article. Updated. Preprint.
References
-
- Bieri, G. , Schroer A. B., and Villeda S. A.. 2023. “Blood‐to‐Brain Communication in Aging and Rejuvenation.” Nature Neuroscience 26: 379–393. - PubMed
-
- Bonardi, C. , de Pulford F., Jennings D., and Pardon M. C.. 2011. “A Detailed Analysis of the Early Context Extinction Deficits Seen in APPswe/PS1dE9 Female Mice and Their Relevance to Preclinical Alzheimer's Disease.” Behavioural Brain Research 222: 89–97. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

