Cortical microstructure is associated with disease severity and clinical progression in genetic frontotemporal dementia: a GENFI study
- PMID: 41068257
- PMCID: PMC12602311
- DOI: 10.1038/s41380-025-03280-x
Cortical microstructure is associated with disease severity and clinical progression in genetic frontotemporal dementia: a GENFI study
Abstract
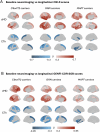
The study of genetic frontotemporal dementia (FTD) allows investigating its earliest presymptomatic stages. Using cross-sectional T1-weighted and diffusion-weighted MRI, we test the hypothesis that cortical microstructural alterations, quantified as cortical mean diffusivity (cMD), are detectable earlier and are more strongly associated with clinical progression than cortical thickness (CTh). The sample comprised n = 710 individuals (47.8 ± 13.5 years, 56.6% female, 14.1 ± 3.3 years of education), including 118 symptomatic carriers and 305 presymptomatic carriers with mutations in C9orf72, GRN or MAPT genes, and 287 non-carriers, collected from 24 GENFI sites. A subset of n = 453 individuals (289 carriers, 164 non-carriers) were investigated across Clinical Dementia Rating (CDR) = 0, 0.5 and ≥1 stages. Two subsets had longitudinal clinical outcome measures, including n = 403 individuals (239 carriers, 164 non-carriers) with Cambridge Behavioural Inventory-Revised scores during 2.8 ± 1.6 years, and n = 261 individuals (164 carriers, 97 non-carriers) with CDR Sum-of-Boxes scores during 2.0 ± 0.8 years. Regional cMD and CTh were entered into linear mixed-effects models incorporating age, sex and education as covariates; site, and individual nested within site were random intercepts. The results demonstrated that cMD is more sensitive than CTh to track early cortical injury, with elevated cMD first observed at CDR = 0 in C9orf72 carriers, followed by MAPT carriers (from CDR = 0.5 stage), and by GRN carriers (beginning at CDR ≥ 1). At all stages, cortical microstructural injury had stronger effect size and was more widespread than cortical thinning. In all mutation carrier types, cMD was more strongly associated than CTh with subsequent clinical progression. Cortical microstructure is a promising biomarker to identify at-risk individuals before atrophy and clinical progression, with utility in therapeutic trials.
© 2025. The Author(s).
Conflict of interest statement
Competing interests: The authors declare no competing interests. Ethical approval and consent to participate: All methods were performed in accordance with the relevant guidelines and regulations, following the Declaration of Helsinki, reviewed and approved by all countries’ respective Ethics committees. All participants signed a written informed consent to participate in the study. This research study was performed in Sweden and approved by the Ethical Review Board (EPN), with reference numbers Dnr 2012/1611-31/1, Dnr 2013/1563-32, Dnr 2017/2097-32, and Dnr 2022-01024-02.
Figures



References
-
- Boeve BF, Boxer AL, Kumfor F, Pijnenburg Y, Rohrer JD. Advances and controversies in frontotemporal dementia: diagnosis, biomarkers, and therapeutic considerations. Lancet Neurol. 2022;21:258–72. - PubMed
-
- Lashley T, Rohrer JD, Mead S, Revesz T. Review: an update on clinical, genetic and pathological aspects of frontotemporal lobar degenerations. Neuropathology Appl Neurobio. 2015;41:858–81. - PubMed
MeSH terms
Substances
Grants and funding
- F2022-0036, F2023-0049, F2024-0064/Neuroförbundet (Swedish Association of Persons with Neurological Disabilities)
- AF-939647, AF-968569, AF-1012445/Alzheimerfonden
- ALF grants FoUI-960437, FoUI-975492/Stockholms Läns Landsting (Stockholm County Council)
- KI Fonder grants/Karolinska Institutet (Karolinska Institute)
- PI21/00791 and PI24/00598/Ministry of Economy and Competitiveness | Instituto de Salud Carlos III (Institute of Health Carlos III)
LinkOut - more resources
Full Text Sources
Miscellaneous

