TRAF2 Promotes Liver Fibrosis via Regulation of the HIF-1α/GLUT1-Mediated Glycolysis in Hepatic Stellate Cells
- PMID: 41079931
- PMCID: PMC12509682
- DOI: 10.7150/ijbs.99682
TRAF2 Promotes Liver Fibrosis via Regulation of the HIF-1α/GLUT1-Mediated Glycolysis in Hepatic Stellate Cells
Abstract
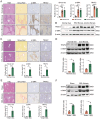
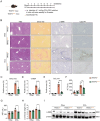
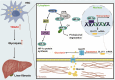
Tumor necrosis factor receptor-associated factor 2 (TRAF2) is an intracellular aptamer protein with E3 ligase activity and has been reported to be involved in the pathogenesis of hepatitis and liver cancer. However, the specific mechanism for liver fibrosis mediated by TRAF2 is a still-unresolved issue. In this study, we uncovered high TRAF2 expression in activated hepatic stellate cells (HSCs) and fibrotic livers of both human and two mouse liver fibrosis models. TRAF2 in HSCs correlated positively with liver fibrosis and could directly prompt HSC activation, as evidenced by in vitro gain-of-function and loss-of-function models. In vivo, HSC-specific knockout of TRAF2 could alleviate liver injury and fibrosis in mice. Mechanistically, we demonstrated that TRAF2 in HSCs promoted the increase of hypoxia-inducible factor-1α (HIF-1α) levels by inhibiting von Hippel-Lindau (pVHL)-mediated HIF-1α degradation and inducing HIF-1α translation via activating mTORC1 pathway. Elevated HIF-1α expression predisposed to a rise in its transcriptional target glucose transporter 1 (GLUT1) expression and glycolytic activity in HSCs, eventually developing liver fibrosis. Thus, TRAF2 exerts a significant impact upon activating HSCs and may become a candidate molecule for anti-liver fibrosis therapy.
Keywords: GLUT1; Glycolysis; HIF-1α; Hepatic stellate cells (HSCs); Liver fibrosis; TRAF2.
© The author(s).
Conflict of interest statement
Competing Interests: The authors have declared that no competing interest exists.
Figures





References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

