The association between serum uric acid and survival outcomes in patients with metastatic renal cell carcinoma treated with targeted therapy
- PMID: 41083961
- PMCID: PMC12519814
- DOI: 10.1186/s12885-025-14962-8
The association between serum uric acid and survival outcomes in patients with metastatic renal cell carcinoma treated with targeted therapy
Abstract
Purpose: Serum uric acid (SUA), the end product of purine metabolism, may play a crucial role in the biology of several cancers, including renal cell carcinoma (RCC). However, data on the association of SUA with metastatic RCC (mRCC) survival are limited. Thus, we evaluated the prognostic importance of baseline serum uric acid in mRCC patients treated with targeted therapy.
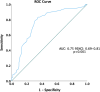
Methods: The SUA level of 5.8 mg/dL was determined as the optimal cut-off point in predicting OS in the receiver operating characteristic curve. The Kaplan-Meier method was used to estimate progression-free survival (PFS) and overall survival (OS) in SUA groups. Cox proportional hazard models were performed to evaluate the association between SUA and survival outcomes.
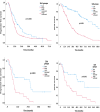
Results: A total of 290 patients with a median age of 61 were included in the study. The median PFS and OS of all patients were 11.6 months and 45 months, respectively. The low-SUA group had superior PFS and OS times than the high-SUA group (16.2 months vs. 8.8 months, p < 0.001 for PFS; 92 months vs. 24.7 months, p < 0.001 for OS, respectively). The multivariate analysis indicated that high SUA levels remained an independent predictor of both PFS and OS after controlling for multiple confounders, including the International Metastatic Renal Cell Carcinoma Database Consortium (IMDC) risk scoring system (HR:1.79, 95%CI 1.35-2.37, p < 0.001 for PFS; HR: 3.32, 95%CI 2.33-4.72, p < 0.001 for OS).
Conclusions: Our study highlights a noteworthy association between high SUA levels and adverse survival outcomes in patients with RCC treated with targeted therapies.
Keywords: Metastatic renal cell carcinoma; Prognosis; Targeted therapy; Uric acid.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: This retrospective study involving human participants was reviewed and approved, and the need for informed consent was waived by the Ethic Commission of Dokuz Eylul University (decision no:2025/20–22). All procedures in the present study were performed in accordance with the 1964 Declaration of Helsinki and its later amendments. Consent for publication: Not applicable. Competing interests: The authors declare no competing interests.
Figures

References
-
- Adhikari A, Sapkota S, Gogia S, Kc O. Changes in the overall survival of patients with metastatic renal cell carcinoma in the era of immune-checkpoint inhibitors. Cancer Epidemiol. 2024;92:102639. 10.1016/j.canep.2024.102639. - PubMed
-
- Heng DYC, Xie W, Regan MM, Warren MA, Golshayan AR, Sahi C, et al. Prognostic factors for overall survival in patients with metastatic renal cell carcinoma treated with vascular endothelial growth factor–targeted agents: results from a large, multicenter study. J Clin Oncol. 2009;27:5794–9. 10.1200/JCO.2008.21.4809. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

