Axon guidance cue SLIT2 regulates the murine skeletal stem cell niche through sympathetic innervation
- PMID: 41090361
- PMCID: PMC12520678
- DOI: 10.1172/JCI193014
Axon guidance cue SLIT2 regulates the murine skeletal stem cell niche through sympathetic innervation
Abstract
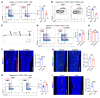
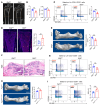
Sympathetic tone is a central signaling axis inhibiting osteogenesis; however, the combination of durable local and systemic sympathetic effects on bone argues that multiple mechanisms, including yet-undiscovered pathways, are involved. Here, we found that sympathetic nerves constituted a component of the skeletal stem cell (SSC) niche: mice with conditional deletion of the classical axonal repellent Slit2 in sympathetic nerves (Slit2th mice), but not in bone stem/progenitor cells or sensory nerves, showed osteopenia due to an increase in sympathetic innervation and an associated decrease in SSCs. Mice with increased skeletal sympathetic innervation displayed impaired SSC niche function in an SSC orthotopic transplantation and engraftment system. Follistatin-like 1 (FSTL1) is a SLIT2-regulated soluble factor suppressing SSC self-renewal and osteogenic capacity. Accordingly, ablation of Fstl1 in sympathetic neurons enhanced SSC-driven osteogenesis and attenuated the bone loss seen in Slit2th mice. Together, the findings indicate that SLIT2 is a regulator of a sympathetic nerve-mediated SSC niche.
Keywords: Bone biology; Bone development; Neuroscience.
Figures




References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

