Oxytocin modulates respiratory heart rate variability through a hypothalamus-brainstem-heart neuronal pathway
- PMID: 41116116
- PMCID: PMC12586189
- DOI: 10.1038/s41593-025-02074-2
Oxytocin modulates respiratory heart rate variability through a hypothalamus-brainstem-heart neuronal pathway
Abstract
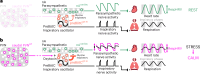
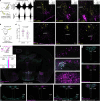
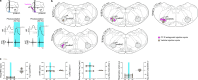
The variation in heart rate in phase with breathing is called respiratory heart rate variability (RespHRV). Relaxation and positive socio-emotional states can amplify RespHRV, yet the underlying mechanism remains largely unknown. Here we identify a hypothalamus-brainstem neuronal pathway in rodents through which oxytocin (OT) amplifies RespHRV during calming behavior. OT neurons from the caudal paraventricular nucleus in the hypothalamus regulate the activity of a subgroup of inhibitory neurons in the pre-Bötzinger complex, the brainstem nucleus that generates the inspiratory rhythm. Specifically, OT enhances the glycinergic input from OT-receptor-expressing neurons in the pre-Bötzinger complex to cardiac-innervating parasympathetic neurons in the nucleus ambiguus during inspiration. This leads to amplified respiratory modulation of parasympathetic activity to the heart, thereby enhancing RespHRV. We show that OT neurons participate in the restoration of RespHRV amplitude during recovery from stress in mice, indicating that OT acts centrally to regulate cardiac activity during a calming behavior.
© 2025. The Author(s).
Conflict of interest statement
Competing interests: The authors declare no competing interests.
Figures















References
-
- Menuet, C. et al. Redefining respiratory sinus arrhythmia as respiratory heart rate variability: an international Expert Recommendation for terminological clarity. Nat. Rev. Cardiol.10.1038/s41569-025-01160-z (2025). - PubMed
-
- Elstad, M., O’Callaghan, E. L., Smith, A. J., Ben-Tal, A. & Ramchandra, R. Cardiorespiratory interactions in humans and animals: rhythms for life. Am. J. Physiol. Heart Circ. Physiol.315, H6–H17 (2018). - PubMed
-
- Fisher, J. P., Zera, T. & Paton, J. F. R. Respiratory–cardiovascular interactions. Handb. Clin. Neurol.188, 279–308 (2022). - PubMed
-
- O’Callaghan, E. L. et al. Enhancing respiratory sinus arrhythmia increases cardiac output in rats with left ventricular dysfunction. J. Physiol.598, 455–471 (2020). - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

