Dimethoxybenzohomoadamantane-based soluble epoxide hydrolase inhibitors: in vivo efficacy in a murine model of chemotherapy-induced neuropathic pain
- PMID: 41128518
- PMCID: PMC12551014
- DOI: 10.1080/14756366.2025.2574990
Dimethoxybenzohomoadamantane-based soluble epoxide hydrolase inhibitors: in vivo efficacy in a murine model of chemotherapy-induced neuropathic pain
Abstract
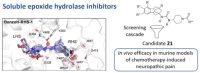
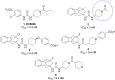
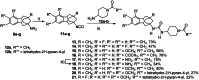
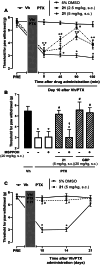
The soluble epoxide hydrolase (sEH) has recently emerged as a promising target for the treatment of several pain-related conditions. Herein, we report the design and synthesis of a peripherally restricted sEH inhibitor with high potency and good Drug Metabolism and Pharmacokinetics (DMPK) properties. Molecular dynamics and X-ray crystallography helped reveal the binding of these inhibitors to sEH. The selected compound showed a robust analgesic effect in a dose-dependent manner in a murine model of chemotherapy-induced neuropathic pain (CINP). Moreover, the compound also prevented the development of paclitaxel-induced neuropathic pain. Overall, these results suggest that peripheral inhibition of sEH might constitute a novel therapy to prevent and treat CINP.
Keywords: Benzohomoadamantane; chemotherapy-induced neuropathic pain; epoxyeicosatrienoic acids; soluble epoxide hydrolase; urea.
Conflict of interest statement
S.V. is a member of the Editorial Board of the Journal of Enzyme Inhibition and Medicinal Chemistry. He was not involved in the assessment, peer review, or decision-making process of this article. S.C., A.L.T., C.G., M.P., C.G.-F. and S.V. are inventors of the Universitat de Barcelona patent application on sEHIs WO2019/243414 and WO2022/200105. C.M. and B.D.H. are inventors of the University of California patents on sEHIs licenced to EicOsis. None of the other authors has any disclosures to declare.
Figures
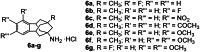
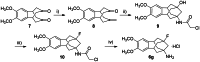


References
-
- Frömel T, Hu J, Fleming I.. Lipid mediators generated by the cytochrome P450-epoxide hydrolase pathway. Adv Pharmacol. 2023;97:327–373. - PubMed
-
- Pillarsetti S, Khanna I.. A multimodal disease modifying approach to treat neuropathic pain—inhibition of soluble epoxide hydrolase (sEH). Drug Discov Today. 2015;20(11):1382–1390. - PubMed
-
- Turnbull J, Chapman V.. Targeting soluble epoxide hydrolase pathway as a novel therapeutic approach for the treatment of pain. Curr Opin Pharmacol. 2024;78:102477. - PubMed
-
- Iyer MR, Kundu B, Wood CM.. Soluble epoxide hydrolase inhibitors: an overview and patent review from the last decade. Expert Opin Ther Pat. 2022;32(6):629–647. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
