Microbial and chemical predictors of methane release from a stratified thermokarst permafrost hotspot
- PMID: 41140401
- PMCID: PMC12549645
- DOI: 10.3389/fmicb.2025.1657143
Microbial and chemical predictors of methane release from a stratified thermokarst permafrost hotspot
Abstract
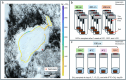
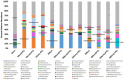
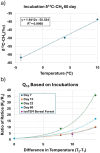
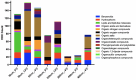
Soils are dynamic interfaces that can act as both sources and sinks of methane (CH₄), yet the microbial processes underlying these fluxes remain poorly constrained in current Earth system models-particularly in thawing permafrost regions. Accurately quantifying subsurface microbial activity and its response to environmental variation is essential for improving predictions of CH₄ emissions under shifting temperature regimes. Here, we explore the potential of volatile organic compounds (VOCs) as early chemical indicators of microbial processes driving CH₄ production within a thermokarst-associated CH₄ hotspot. Field surveys at Big Trail Lake, a young thermokarst feature in central Alaska, identified localized CH₄ emission zones. Anaerobic soil laboratory microcosms from 50, 200, and 400 cm depths were incubated at -4 °C, 5 °C, and 12 °C to simulate freeze-thaw transitions. Methane flux increased markedly with temperature, and microbial community shifts revealed Methanosarcina spp. as the dominant methanogen, particularly at 200 cm. VOC profiling showed strong depth- and temperature-dependent patterns, with the 50 cm layer exhibiting the greatest chemical diversity. Notably, 200 cm soils produced VOC signatures overlapping with those from pure Methanosarcina acetivorans C2A cultures, supporting the identification of shared metabolites linked to active methanogenesis. Extended 60-day incubations confirmed temperature-sensitive CH₄ production. Carbon isotopic enrichment in CH₄ was unexpectedly strong with warming, and metagenomic detection of ANME-associated markers-including multiheme cytochromes and formate dehydrogenases-supports temperature-sensitive anaerobic oxidation of methane as a significant control on isotopic signatures. Calculated Q₁₀ values for methanogenesis exceeded typical values for boreal soils, highlighting an underappreciated temperature responsiveness of Arctic methanogens. Together, these results demonstrate that VOCs can serve as informative biomarkers of subsurface microbial activation and offer a novel diagnostic tool for detecting early-stage CH₄ hotspot formation. Incorporating such chemically and biologically resolved metrics into process-based models will be critical for improving forecasts of CH₄ release from thawing permafrost landscapes.
Keywords: biogeochemical modeling; carbon cycling; methane hotspot; methane isotopes; methanotrophy; microbial VOCs; permafrost thaw; thermokarst soils.
Copyright © 2025 Rozmiarek, Yang, Schambach, Bennett, Caro, Sammon, Whiting, Miller, Ricken, Bigler, Jayne, Fukuyama, Jones and Smallwood.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures




References
-
- Albers C. N., Kramshøj M., Rinnan R. (2018). Rapid mineralization of biogenic volatile organic compounds in temperate and Arctic soils. Biogeosciences 15, 3591–3601. doi: 10.5194/bg-15-3591-2018 - DOI
-
- Bar-Or I., Elvert M., Eckert W., Kushmaro A., Vigderovich H., Zhu Q., et al. (2017). Iron-coupled anaerobic oxidation of methane performed by a mixed bacterial-archaeal community based on poorly reactive minerals. Environ. Sci. Technol. 51, 12293–12301. doi: 10.1021/acs.est.7b03126, PMID: - DOI - PubMed
-
- Basu S., Lan X., Dlugokencky E., Michel S., Schwietzke S., Miller J. B., et al. (2022). Estimating emissions of methane consistent with atmospheric measurements of methane and δ13C of methane. Atmos. Chem. Phys. 22, 15351–15377. doi: 10.5194/acp-22-15351-2022 - DOI
LinkOut - more resources
Full Text Sources
Miscellaneous

