Combined Adaptive Immune Mechanisms Mediate Cardiac Injury After COVID-19 Vaccination
- PMID: 41164857
- PMCID: PMC12643570
- DOI: 10.1161/CIRCULATIONAHA.125.074644
Combined Adaptive Immune Mechanisms Mediate Cardiac Injury After COVID-19 Vaccination
Abstract
Background: The COVID-19 pandemic, caused by SARS-CoV-2, has led to the first approval of mRNA vaccines in humans. By producing the full-length SARS-CoV-2 Spike protein, they induce protective antiviral immunity. Acute myopericarditis (AMP) development after vaccination has repeatedly been reported; however, the pathogenesis of this complication remains elusive.
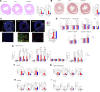
Methods: In-depth phenotyping of peripheral blood T cells was undertaken in cohorts of patients who developed AMP after mRNA vaccination, patients hospitalized for severe COVID-19, and healthy subjects with no cardiac side effects after mRNA vaccine. Validation studies were carried out using an experimental model of cardiac inflammation, in which a shared epitope elicits functional responses in patients and mice and induces AMP.
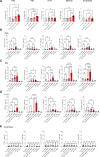
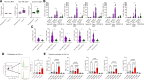
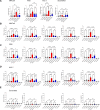
Results: We show that T cells from patients with AMP recognize vaccine-encoded Spike epitopes homologous to those of cardiac self-proteins. One of these epitopes, mimicking an amino acid sequence from a cardiomyocyte-expressed K+ channel, induced AMP in mice. When functional responses to the Kv2 were analyzed, patients with AMP after mRNA vaccination, but not patients with COVID-19, displayed an expanded pattern of cytokine production similar to that observed in AMP mice and in autoimmune myocarditis. Crucially, T-cell autoimmunity segregates to cardiotropic cMet (c-mesenchymal epithelial transition factor)-expressing T cells and is prevented by cMet inhibition, suggesting that heart homing imprinting, permitted by the unique mRNA vaccine biodistribution, is required for AMP development.
Conclusions: AMP development after mRNA vaccines is mediated by distinct immune components, including molecular mimicry, T-cell receptor affinity, and, importantly, homing imprinting.
Keywords: COVID-19; T-lymphocytes; autoimmunity; vaccination.
Conflict of interest statement
Dr Marelli-Berg is a consultant for AstraZeneca.
Figures



References
-
- Park JK, Lee EB, Winthrop KL. What rheumatologists need to know about mRNA vaccines: current status and future of mRNA vaccines in autoimmune inflammatory rheumatic diseases. Ann Rheum Dis. 2024;83:687–695. doi: 10.1136/ard-2024-225492 - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

