The Impact of Immune-Related Adverse Events on the Survival of Patients Treated with Immune Checkpoint Inhibitors: The Distinct Role of Cardiac Toxicities
- PMID: 41227189
- PMCID: PMC12609892
- DOI: 10.3390/jcm14217794
The Impact of Immune-Related Adverse Events on the Survival of Patients Treated with Immune Checkpoint Inhibitors: The Distinct Role of Cardiac Toxicities
Abstract
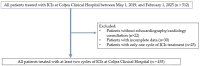
Background: Immunotherapy with immune checkpoint inhibitors (ICIs) has fundamentally transformed cancer treatments. Unfortunately, its benefits are accompanied by the occurrence of immune-related adverse events (irAEs). While non-cardiac irAEs have been consistently associated with a favorable prognosis, the impact of cardiac toxicities remains insufficiently explored. Methods: We conducted a retrospective, observational study at the Oncology Department of Colțea Clinical Hospital, Bucharest. All the patients treated with ICIs between 1 May 2019 and 1 February 2024 were selected in the initial cohort. Of 512 eligible patients, 435 were included in the final analysis, with comprehensive recordings of clinical, oncological, and cardiac monitoring parameters, and at least one complete cycle of ICI treatment. Adverse events were classified according to CTCAE v5.0, and overall survival (OS) and progression-free survival (PFS) were assessed using Kaplan-Meier estimates and Cox regression models. Results: Our results showed that patients who developed non-cardiac irAEs experienced a significant survival benefit: median OS 26.0 months (95% CI, 15.5-NA) vs. 13.9 months (95% CI, 12.4-16.5), 0.66 (95% CI, 0.49-0.9) hazard ratio (HR); median PFS 12.3 months (95% CI, 8.1-26.0) vs. 8.7 months (95% CI, 7.3-10.3), 0.74 (95% CI, 0.56-0.97) HR. Conversely, patients with cardiac toxicities did not derive the same advantage, with similar OS and PFS values that did not reach statistical significance: median OS 15.0 months (95% CI, 13.3-19.3) vs. 15.8 months (95% CI, 12.0-30.3), 1.11 (95% CI, 0.78-1.57) HR; median PFS 9.1 months (95% CI, 7.6-10.4) vs. 8.1 months (95% CI, 5.3-19.3), 1.003 (95% CI, 0.72-1.39) HR. Conclusions: These findings support the role of non-cardiac irAEs as markers of favorable therapeutic response, while cardiac irAEs do not confer the same prognostic benefit. The results underscore the importance of active cardiovascular monitoring and close multidisciplinary collaboration in the management of patients receiving ICIs.
Keywords: cardiotoxicity; immune checkpoint inhibitors; immune-related adverse events; progression-free survival; real-world data; survival analysis.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

References
-
- Nardin S., Ruffilli B., Costantini P., Mollace R., Taglialatela I., Pagnesi M., Chiarito M., Soldato D., Cao D., Conte B., et al. Navigating Cardiotoxicity in Immune Checkpoint Inhibitors: From Diagnosis to Long-Term Management. J. Cardiovasc. Dev. Dis. 2025;12:270. doi: 10.3390/jcdd12070270. - DOI - PMC - PubMed
-
- Cook S., Samuel V., Meyers D.E., Stukalin I., Litt I., Sangha R., Morris D.G., Heng D.Y.C., Pabani A., Dean M., et al. Immune-Related Adverse Events and Survival Among Patients With Metastatic NSCLC Treated With Immune Checkpoint Inhibitors. JAMA Netw. Open. 2024;7:e2352302. doi: 10.1001/jamanetworkopen.2023.52302. - DOI - PMC - PubMed
-
- Hussaini S., Chehade R., Boldt R.G., Raphael J., Blanchette P., Maleki Vareki S., Fernandes R. Association between Immune-Related Side Effects and Efficacy and Benefit of Immune Checkpoint Inhibitors—A Systematic Review and Meta-Analysis. Cancer Treat. Rev. 2021;92:102134. doi: 10.1016/j.ctrv.2020.102134. - DOI - PubMed
LinkOut - more resources
Full Text Sources

