Cost-effectiveness of introducing a maternal vaccine or long-acting monoclonal antibody to prevent infant respiratory syncytial virus disease in Nepal
- PMID: 41232121
- PMCID: PMC12615004
- DOI: 10.7189/jogh.15.04292
Cost-effectiveness of introducing a maternal vaccine or long-acting monoclonal antibody to prevent infant respiratory syncytial virus disease in Nepal
Abstract
Background: The World Health Organization recommends two passive immunisation strategies to prevent respiratory syncytial virus (RSV) disease in young infants. Both are being introduced in high-income settings, but their affordability and cost-effectiveness have not been evaluated in many low- and middle-income countries. Preliminary estimates of cost-effectiveness are needed to guide immunisation policy and planning in Nepal.
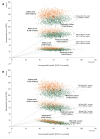
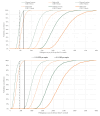
Methods: We estimated the potential health impact and cost-effectiveness of introducing a maternal vaccine (RSVpreF) or long-acting infant monoclonal antibody (mAb) (nirsevimab) over the period 2025-34 in Nepal. We compared both interventions to the status quo (no intervention) and to each other. Model inputs included health care cost estimates from a recent prospective cost-of-illness study in Kathmandu, as well as the latest efficacy data from clinical trials. The primary outcome measure was the incremental cost (2023 USD) per disability-adjusted life year (DALY) averted from a governmental health perspective. We conducted a range of deterministic analyses, including scenarios that incorporated a societal perspective and a seasonal approach. Additionally, we performed probabilistic uncertainty analyses to assess decision uncertainty and estimated the likelihood of cost-effectiveness for each intervention across a range of willingness-to-pay thresholds.
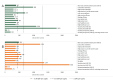
Results: Introducing a maternal vaccine (USD 5/dose, 81% coverage, 69% efficacy, 6 months protection) or long-acting infant mAb (USD 5/dose, 97% coverage, 77% efficacy, 5 months protection) could prevent >2300 deaths and >50 000 hospital admissions over ten years. The discounted immunisation programme costs were estimated to be USD 30 and USD 35 million, respectively. Compared to the status quo, the maternal vaccine and the long-acting infant mAb were estimated to cost USD 387 and USD 486 per DALY averted, respectively, which is around 0.3 times and 0.4 times the national gross domestic product (GDP) per capita. There was a 95% probability that the maternal vaccine would be cost-effective at USD 5 per dose, assuming a willingness-to-pay threshold of 0.5 times the national GDP per capita. With our base case assumptions, the maternal vaccine dominated the mAb (i.e. generated more health benefits at a lower cost). However, the results (and the rank order of interventions) were sensitive to the dose price, efficacy, duration of protection, and RSV disease burden estimates. Cost-effectiveness of the mAb improves with timely administration or when a seasonal approach is implemented.
Conclusions: New passive immunisation strategies have the potential to prevent a substantial number of RSV-related hospitalisations and deaths in Nepal. Cost-effectiveness and product choice will heavily depend on the price negotiated for each product.
Copyright © 2025 by the Journal of Global Health. All rights reserved.
Conflict of interest statement
Disclosure of interest: The authors completed the ICMJE Disclosure of Interest Form (available upon request from the corresponding author) and declare the following activities and relationships: Louis J Bont has regular interaction with pharmaceutical and other industrial partners and has not received personal fees or other personal benefits. Louis J Bont is the founding chair of the Respiratory Syncytial Virus Foundation. The University Medical Centre Utrecht (UMCU) has received significant funding (>EUR 100 000 per industrial partner) for investigator-initiated studies from AbbVie, MedImmune, Janssen Pharmaceuticals, the Bill & Melinda Gates Foundation, Nutricia (Danone), and MeMed Diagnostics. UMCU has received significant cash or in-kind funding as part of the public-private partnership Innovative Medicines Initiative – funded Respiratory Syncytial Virus Consortium in Europe project, from GlaxoSmithKline, Novavax, Janssen, AstraZeneca, Pfizer, and Sanofi. UMCU has received major funding from Julius Clinical for participating in the International Network for Optimal Resistance Monitoring of RSV study sponsored by MedImmune. UMCU has received minor funding for participation in trials by Regeneron Pharmaceuticals and Janssen from 2015 to 2017 (total annual estimate <EUR 20 000). UMCU received minor funding for consultation and invited lectures by AbbVie, MedImmune, Ablynx, Bavarian Nordic, mAbxience, Novavax, Pfizer, and Janssen (total annual estimate <EUR 20 000). PATH receives funding from the Bill & Melinda Gates Foundation to support its work related to RSV.
Figures
References
-
- Li Y, Wang X, Blau DM, Caballero MT, Feikin DR, Gill CJ, et al. Global, regional, and national disease burden estimates of acute lower respiratory infections due to respiratory syncytial virus in children younger than 5 years in 2019: a systematic analysis. Lancet. 2022;399:2047–64. 10.1016/S0140-6736(22)00478-0 - DOI - PMC - PubMed
-
- Zhang S, Akmar LZ, Bailey F, Rath BA, Alchikh M, Schweiger B, et al. Cost of Respiratory Syncytial Virus-Associated Acute Lower Respiratory Infection Management in Young Children at the Regional and Global Level: A Systematic Review and Meta-Analysis. J Infect Dis. 2020;222:S680–7. 10.1093/infdis/jiz683 - DOI - PubMed
-
- World Health Organization. WHO position paper on immunization to protect infants against respiratory syncytial virus disease, May 2025. Geneva, Switzerland: World Health Organization; 2025. Available: https://iris.who.int/server/api/core/bitstreams/96ec533a-de56-4e1a-8e01-.... Accessed: 6 September 2025.
-
- Simões EAF, Madhi SA, Muller WJ, Atanasova V, Bosheva M, Cabañas F, et al. Efficacy of nirsevimab against respiratory syncytial virus lower respiratory tract infections in preterm and term infants, and pharmacokinetic extrapolation to infants with congenital heart disease and chronic lung disease: a pooled analysis of randomised controlled trials. Lancet Child Adolesc Health. 2023;7:180–9. 10.1016/S2352-4642(22)00321-2 - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
