A Multicomponent Intervention to Improve Maternal Infection Outcomes
- PMID: 41259754
- PMCID: PMC7618407
- DOI: 10.1056/NEJMoa2512698
A Multicomponent Intervention to Improve Maternal Infection Outcomes
Abstract
Background: Maternal infection and sepsis are major causes of maternal death and severe illness worldwide, particularly in low- and middle-income countries. Inconsistent implementation of evidence-based recommendations for infection prevention and management and delays in detection and treatment of maternal sepsis contribute to the number of preventable deaths.
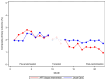
Methods: We conducted a cluster-randomized trial to assess a multicomponent intervention, the Active Prevention and Treatment of Maternal Sepsis (APT-Sepsis) program. This program was designed to support health care providers in achieving three goals: adherence to World Health Organization (WHO) hand-hygiene standards; adoption of evidence-based practices for maternal infection prevention and management; and early detection of sepsis and use of the FAST-M (fluids, antibiotics, source control, transfer if required, and monitoring) treatment bundle. Usual care was provided in the control group, along with dissemination of guidelines. The primary outcome was a composite of infection-related maternal death, infection-related near-miss event (events in which women survived a life-threatening complication), or severe infection-related illness (deep surgical-site, deep perineal, or body-cavity infection) among women who were pregnant or had recently been pregnant.
Results: We randomly assigned 59 health facilities (where 431,394 women gave birth during the trial) in Malawi and Uganda to the intervention group (30 clusters) or the usual-care group (29 clusters). A primary-outcome event occurred in 1.4% of the patients in the intervention group and in 1.9% of those in the usual-care group (risk ratio, 0.68; 95% confidence interval, 0.55 to 0.83; P<0.001). This effect was generally consistent between countries and among facilities of difference sizes and was sustained over time.
Conclusions: Implementation of the APT-Sepsis program led to a significantly lower risk of a composite of infection-related maternal death, infection-related near-miss event, or severe infection-related illness than usual care. (Funded by the Joint Global Health Trials scheme and others; APT-Sepsis ISRCTN number, ISRCTN42347014.).
Copyright © 2025 Massachusetts Medical Society.
Figures


References
-
- James SL, Abate D, Abate KH, et al. Global, regional, and national incidence, prevalence, and years lived with disability for 354 diseases and injuries for 195 countries and territories, 1990–2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet. 2018;392:1789–858. doi: 10.1016/S0140-6736(18)32279-7. - DOI - PMC - PubMed
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
