Characteristics of KRAS WT pancreatic adenocarcinomas: results of a large French multicentric cohort
- PMID: 41262210
- PMCID: PMC12623649
- DOI: 10.1177/17588359251339939
Characteristics of KRAS WT pancreatic adenocarcinomas: results of a large French multicentric cohort
Abstract
Background: Genome and transcriptome analysis has enhanced the characterisation of pancreatic ductal adenocarcinoma (PDAC), paving the way for targeted therapies. Tumours KRAS wild type (WT) represent a unique subgroup.
Objectives: Characterise the population and molecular abnormalities present in KRAS WT PDAC.
Design: Clinical and molecular data from a large retrospective cohort of KRAS WT PDAC were analysed.
Methods: Next-generation sequencing (NGS) was used to analyse DNAs and RNAs, allowing molecular and transcriptomic characterisation.
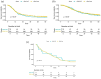
Results: We identified 93/1059 (9%) KRAS WT PDAC, among which eight had druggable fusions (n = 8/30 contributive samples), six had BRAF mutations and 19 (n = 19/47) had mutations in homologous recombination (HR) pathway genes. Potential molecular targets in this series may be underestimated due to many non-contributive results. Clinical characteristics and survival did not differ between patients with KRAS WT and KRAS-mutated tumours. Transcriptomic data were available for 350 samples. Their analysis shows a difference in phenotype between mutated and WT tumours, with a molecular profile that appears to be better prognostic for KRAS WT tumours.
Conclusion: KRAS WT tumours are enriched with molecular abnormalities of therapeutic interest. These include oncogene driver alterations (gene fusions and mutations) and mutations in genes of the HR pathway. Targeted therapy strategies for PDAC rely on molecular testing beyond RAS, but further research is needed to identify new therapeutic approaches that improve outcomes in PDAC.
Keywords: KRAS mutation; genomics; molecular profiling; next-generation sequencing; pancreatic cancer.
© The Author(s), 2025.
Conflict of interest statement
P.L.-P. received honorarium from Biocartis, Amgen, Pierre Fabre Servier for consulting. J.-B.B. received personal fees from Abbvie, Amgen, Bayer, Bristol Myers Squibb, GSK, Merck Serono, Merck Sharp & Dohme, Pierre Fabre, Sanofi, Servier, Takeda and non-financial support from Amgen, Merck Serono and Roche, outside the submitted work.
Figures


References
-
- Rahib L, Smith BD, Aizenberg R, et al. Projecting cancer incidence and deaths to 2030: the unexpected burden of thyroid, liver, and pancreas cancers in the United States. Cancer Res 2014; 74(11): 2913–2921. - PubMed
-
- Siegel RL, Miller KD, Fuchs HE, et al. Cancer statistics, 2021. CA Cancer J Clin 2021; 71(1): 7–33. - PubMed
-
- Conroy T, Desseigne F, Ychou M, et al. FOLFIRINOX versus gemcitabine for metastatic pancreatic cancer. N Engl J Med 2011; 364(19): 1817–1825. - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

