Shape analysis of the amygdala, hippocampus and thalamus in former American football players
- PMID: 41277896
- PMCID: PMC12631119
- DOI: 10.1093/braincomms/fcaf440
Shape analysis of the amygdala, hippocampus and thalamus in former American football players
Abstract
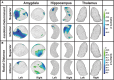
Repetitive head impacts are common in contact and collision sports and are linked to structural brain changes and an elevated risk of neurodegenerative diseases such as Chronic Traumatic Encephalopathy. Identifying early in vivo structural markers remains challenging. Although diagnosis currently requires post-mortem confirmation, clinical symptoms, including cognitive impairment and behavioural changes, are reflected in the diagnosis of Traumatic Encephalopathy Syndrome. These symptoms align with dysfunction in key brain regions-amygdala, hippocampus and thalamus-which support memory, emotion and behaviour and commonly show tau pathology in Chronic Traumatic Encephalopathy. This study uses shape analysis to examine structural differences in these regions between former American football players and unexposed asymptomatic controls and evaluates the influence of age, head impact exposure and clinical diagnosis on brain structure. We analyzed brain morphology in former American football players (n = 163) and unexposed, asymptomatic controls (n = 53). Structural segmentation was performed with FreeSurfer 7.1, and the shape analysis pipeline was used to generate subregional reconstructions. Vertex-level morphometry, based on the logarithm of the Jacobian determinant and radial distance, quantified local surface area dilation and thickness. Group differences were examined with covariate-adjusted linear regression models contrasting football players and controls, as well as participants with and without a Traumatic Encephalopathy Syndrome diagnosis. Partial correlations examined the influence of age, age of first football exposure and cumulative head impact index metrics, including frequency, linear acceleration and rotational force. Models were adjusted accordingly for age, body mass index, education, race, imaging site, apolipoprotein status and total intracranial volume. Former football players exhibited bilateral surface area contractions in the hippocampus and amygdala, along with reduced amygdala thickness, compared to controls. Older age was associated with widespread surface contractions and thinning across all regions, except for preserved thickness in the left hippocampus. An earlier age of first exposure to football correlated with surface contractions in the thalamus and left hippocampus. Greater cumulative linear acceleration was linked to bilateral hippocampal surface contractions and reduced thickness in the left thalamus, while greater rotational force exposure was associated with hippocampal thinning. No significant structural differences were found between players with and without a diagnosis of Traumatic Encephalopathy Syndrome. These findings extend volume-based research by revealing localized alterations in surface area dilation and thickness and emphasize the roles of age and repetitive head impact exposure in long-term brain changes.
Keywords: neuroimaging; repetitive head impacts; shape analysis; sports-related head injury; structural MRI.
© The Author(s) 2025. Published by Oxford University Press on behalf of the Guarantors of Brain.
Conflict of interest statement
L.J.B. is the Editor-in-Chief of the Journal of Neuro-Ophthalmology. C.B. receives research support from the Ultimate Fighting Championship, Top Rank promotions and Haymon Boxing. A.P.L. has an ownership interest as a Co-founder of BrainSpec and consults for Agios Pharmaceuticals, Biomarin Pharmaceuticals, DesignTx and Moncon MRI. J.L.C. has provided consultation to Acadia, Actinogen, Acumen, AlphaCognition, Aprinoia, AriBio, Artery, Biogen, BioVie, Bristol-Myers Squib, Cassava, Cerecin, Diadem, EIP Pharma, Eisai, GemVax, Genentech, GAP Innovations, Janssen, Jocasta, Karuna, Lighthouse, Lilly, Lundbeck, LSP/EQT, Merck, NervGen, Novo Nordisk, Oligomerix, Optoceutics, Ono, Otsuka, PRODEO, Prothena, ReMYND, Roche, Sage Therapeutics, Signant Health, Simcere, Suven, SynapseBio, TrueBinding, Vaxxinity, and Wren pharmaceutical, assessment, and investment companies. He owns the copyright of the Neuropsychiatric Inventory. Dr. Eric M. Reiman is a compensated scientific advisor for Alkahest, Alzheon, Aural Analytics, Denali, Green Valley, Retromer Therapeutics, and Vaxxinity, and a co-founder of ALZPath. R.A.S. is a member of the Board of Directors of King-Devick Technologies, Inc. (Chicago, IL, USA), and he receives royalties for published neuropsychological tests from Psychological Assessment Resources, Inc. (Lutz, FL, USA).
Figures






References
-
- Alosco ML, Tripodis Y, Rowland B, et al. A magnetic resonance spectroscopy investigation in symptomatic former NFL players. Brain Imaging Behav. 2019;14:1419–1429.
Grants and funding
LinkOut - more resources
Full Text Sources
