Patient-reported outcomes after ipsilateral radiation therapy for N2b tonsillar squamous cell carcinoma
- PMID: 41288848
- PMCID: PMC12647442
- DOI: 10.1007/s12672-025-03896-z
Patient-reported outcomes after ipsilateral radiation therapy for N2b tonsillar squamous cell carcinoma
Abstract
Background: Previous studies have reported excellent disease control and survival in patients with well-lateralized, American Joint Committee on Cancer (AJCC)-7 T1-2N2b tonsillar squamous cell carcinoma (SCC). The reduced treatment volume is associated with lower rates of physician-assessed toxicity. Patient-reported outcomes (PROs) have been proposed as a similarly reliable measure, but the body of literature is limited for unilaterally treated patients. Our goal was to review PROs of such patients who had reduced treatment volumes.
Methods: We reviewed PROs of patients with AJCC-7 T1-2N2b disease treated with ipsilateral radiation therapy (RT), with or without surgery or chemotherapy before RT. PROs were measured using the MD Anderson Symptom Inventory for head and neck cancer after a median of 28.9 months.
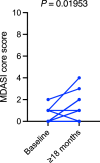
Results: Forty-eight patients were included in the study: 36 (75%) had human papillomavirus-positive disease, 20 (42%) had ≥ 2 nodal levels involved, 15 (31%) had extranodal extension and all patients had N2b disease. Most patients reported no symptoms; a median 72.9% for all 10 head-and-neck symptoms, 75.0% for all 12 core symptoms, and 83.3% for all 6 interference symptoms reported a score of “0”. The most common head-and-neck and core symptoms were dry mouth (n = 33, 69%) and fatigue (n = 23, 48%). Treatment mostly interfered with general activities. PROs were not affected by receipt of surgery; however, receipt of systemic therapy was associated with worse general activity (P = 0.044). Longitudinal analyses revealed mildly worse dry mouth (from 0 of 10 points to 1.5 of 10, P = 0.012) and numbness/tingling (from 0 of 10 to 0.5 of 10, P = 0.020).
Conclusions: Ipsilateral neck RT for N2b tonsil SCC was associated with only a mild PRO symptom burden after at least 18 months, and therefore, a well-tolerated toxicity reduction strategy for appropriate patients.
Keywords: HPV; MD Anderson symptom inventory for head and neck; MDADI; MDASI; PRO; Toxicity; Unilateral radiation therapy; Xerostomia.
Conflict of interest statement
Declarations. Ethics approval and consent to participate: This study was conducted according to the guidelines of the Declaration of Helsinki and was approved by The University of Texas MD Anderson Cancer Center Institutional Review Board for HN IMRT protocol PA2014-0194. Informed consent was obtained from all subjects involved in the study. Consent for publication: Not applicable. Competing interests: The authors declare no competing interests.
Figures



References
-
- Dirix P, Nuyts S, Van den Bogaert W. Radiation-induced xerostomia in patients with head and neck cancer: a literature review. Cancer. 2006;107(11):2525–34. - PubMed
-
- Jereczek-Fossa BA, Santoro L, Alterio D, Franchi B, Fiore MR, Fossati P, et al. Fatigue during head-and-neck radiotherapy: prospective study on 117 consecutive patients. Int J Radiat Oncol Biol Phys. 2007;68(2):403–15. - PubMed
-
- Murphy BA, Gilbert J, Ridner SH. Systemic and global toxicities of head and neck treatment. Expert Rev Anticancer Ther. 2007;7(7):1043–53. - PubMed
-
- Langendijk JA, Doornaert P, Verdonck-de Leeuw IM, Leemans CR, Aaronson NK, Slotman BJ. Impact of late treatment-related toxicity on quality of life among patients with head and neck cancer treated with radiotherapy. J Clin Oncol. 2008;26(22):3770–6. - PubMed
