Emergence of Acquired HIV Drug Resistance Among Individuals Receiving Dolutegravir-Based Antiretroviral Therapy in Uganda: A National Laboratory-Based Survey 2023
- PMID: 41311912
- PMCID: PMC12651561
- DOI: 10.1093/ofid/ofaf679
Emergence of Acquired HIV Drug Resistance Among Individuals Receiving Dolutegravir-Based Antiretroviral Therapy in Uganda: A National Laboratory-Based Survey 2023
Abstract
Background: As dolutegravir (DTG)-based regimens (DBRs) become more widely used and patients remain on treatment longer, cases of virological failure remain rare. This study presents findings from the second annual round of acquired HIV drug resistance (HIVDR) surveillance in Uganda, among individuals with viral nonsuppression (≥1000 copies/mL) receiving DBRs for ≥9 months. The first round was conducted from February to April 2022.
Methods: This nationally representative cross-sectional survey analyzed randomly selected remnant plasma and dried blood spot specimens collected between April and July 2023 from the Central Public Health Laboratories. Genotyping targeted the integrase, protease, and reverse transcriptase regions of the HIV-1 pol gene, resistance was analyzed using the Stanford HIVDR Database. Weighted HIVDR prevalence and 95% confidence intervals (CIs) were calculated for children (0-14 years) and adults (≥15 years).
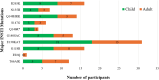
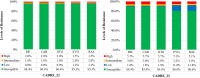
Results: Out of 857 specimens tested, 400 (46.7%) were from children and 457 (53.3%) from adults. Median ages were 11 years for children and 36 years for adults. Median time on DBRs was 1.9 years for children and 2.4 years for adults. Five hundred and fifty-nine (65.2%) specimens were successfully genotyped. The prevalence of DTG resistance was 10.1% (95% CI: 6.4%-13.9%) in children and 8.6% (95% CI: 3.9%-13.3%) in adult, higher than the first round with a prevalence of 6.6% (95% CI: 3.5%-9.6%) and 4.4% (95% CI: 0.7%-7.1%), respectively.
Conclusions: The increase in DTG resistance among both children and adults highlights the need to strengthen adherence and enhance early identification of individuals at risk of HIVDR through novel and existing programmatic interventions.
Keywords: CADRE; Uganda; acquired resistance to dolutegravir.
© The Author(s) 2025. Published by Oxford University Press on behalf of Infectious Diseases Society of America.
Conflict of interest statement
Potential conflicts of interest. The authors do not have any conflict of interest relevant to this work. Financial support. This work was supported by the US President's Emergency Plan for AIDS Relief (PEPFAR) through the Centers for Disease Control and Prevention under the terms of the cooperative agreement, award number 5NU2GGH002132-03. Data availability. The sequence data supporting the findings of this study have been deposited in GenBank, and the accession numbers are PV988491–PV989523. Author contributions. Conceived and designed the study: P. K., J. N. A., C. W., J. d. F. d. S., G. N., D. S., G. S., M. Nan., H. N., M. Nab., U. K. K., R. B., M. S., C. K., E. N. M., H. B., E. N., S. P., E. R.. D. P. Z., C. Z., S. H., M. Nal., and E. K. M. Performed the study: P. K., J. N. A., C. W., G. N., D. S., G. S., M. Nan., U. K. K., R. B., and H. N. Analyzed the data: S. P., J. N. A., C. W., D. S., G. S., M. Nan., and P. K. Wrote the paper: J. N. A., C. W., J. d. F. d. S., G. N., D. S., G. S., M. Nan, H. N., M. Nab., M. S., C. K., E. N. M., H. B., E. N., S. P., E. R., D. P. Z., C. Z., S. H., M. Nal., and P. K.
Figures
References
-
- UNAIDS . UNAIDS Global AIDS Strategy 2021–2026. https://www.unaids.org/en/resources/documents/2021/2021-2026-global-AIDS....
-
- Llibre JM, Pulido F, García F, García Deltoro M, Blanco JL, Delgado R. Genetic barrier to resistance for dolutegravir. AIDS Rev 2015; 17:56–64.. - PubMed
-
- World Health Organization . Policy brief: update of recommendations on first-and second-line antiretroviral regimens. World Health Organization, 2019. https://www.who.int/publications/i/item/WHO-CDS-HIV-19.15.
