Computer image modeling of pentamer packing in polyoma virus "hexamer" tubes
- PMID: 6089394
- PMCID: PMC4167661
- DOI: 10.1016/0304-3991(84)90064-0
Computer image modeling of pentamer packing in polyoma virus "hexamer" tubes
Abstract
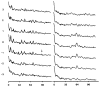
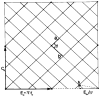
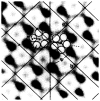
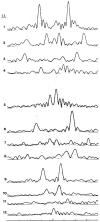
Polymorphic assemblies of polyoma virus capsomeres that have been called "hexamer" tubes (because the morphological units are six-coordinated) are, in fact, built of pentamers, as is the icosahedrally symmetric T = 7d virus capsid. We have established the pentameric form of the capsomeres in the "hexamer" tubes by analysis of low-irradiation micrographs. Methods for generating computer image models with adjustable parameters have been developed to fit micrographs of negatively stained, flattened tubes. The image model has been refined to define the packing arrangement and substructure of the pentametric capsomeres in the superimposed top and bottom layers of the tube and to represent the differential flattening, lateral distortion and staining of the two sides. Information about the structure that is not directly accessible by conventional image filtering methods can be obtained by image modeling methods.
Figures