Hindered depolarizing rotations of perylene in lipid bilayers. Detection by lifetime-resolved fluorescence anisotropy measurements
- PMID: 7356968
- PMCID: PMC6941142
- DOI: 10.1021/bi00546a013
Hindered depolarizing rotations of perylene in lipid bilayers. Detection by lifetime-resolved fluorescence anisotropy measurements
Abstract
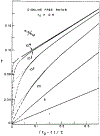
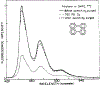
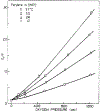
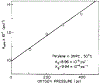
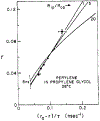
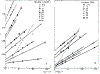
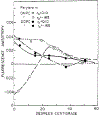
Oxygen quenching of perylene fluorescence was used to vary its fluorescence lifetime. Steady-state fluorescence anisotropy measurements under these quenching conditions were used to investigate the diffusive motions of perylene in the isotropic solvent propylene glycol and in lipid bilayers. These lifetime-resolved anisotropy measurements indicate that the anisotropy of perylene in propylene glycol decays to zero at times long compared to its fluorescence lifetime. In contrast, the asymptotic or "limiting" anistropy values at these long times (r infinity) are nonzero in vesicles of dimyristoyl-phosphatidylcholine (DMPC). r infinity values are largest at temperatures below the DMPC phase transition temperature of 23 degrees C. Representative values of r infinity for perylene in DMPC vesicles are 0.16 and 0.02 at 5 and 47 degrees C, respectively. Thus, in contrast to the free rotations observed for perylene in propylene glycol, perylene rotations are hindered in lipid bilayers. Less marked, yet significant, rotational hindrance was observed in dioleoylphosphatidylcholine (DOPC) vesicles. Representative values for r infinity in this unsaturated lipid are 0.05 and 0.01 at 2 and 45 degrees C, respectively. Steady-state anisotropy measurements with short-wavelength excitations were used to investigate whether the in-plane or out-of-plane rotations of perylene were responsible for the observed r infinity values. In DMPC vesicles we conclude that both rotations are partially hindered. In DOPC vesicles we can only conclude that one or both of these rotations are partially hindered, but both are not free. Most importantly, the existence of fundamentally different diffusive behavior for perylene in solvents and in lipids calls into question the meaning of membrane microviscosities which are derived via such comparisons.
Figures



Similar articles
-
Differential polarized phase fluorometric investigations of diphenylhexatriene in lipid bilayers. Quantitation of hindered depolarizing rotations.Biochemistry. 1979 Feb 6;18(3):508-19. doi: 10.1021/bi00570a021. Biochemistry. 1979. PMID: 420797 Free PMC article.
-
Detection of hindered rotations of 1,6-diphenyl-1,3,5-hexatriene in lipid bilayers by differential polarized phase fluorometry.Biophys J. 1978 Oct;24(1):213-31. doi: 10.1016/S0006-3495(78)85357-0. Biophys J. 1978. PMID: 708824 Free PMC article.
-
Quantitation of hindered rotations of diphenylhexatriene in lipid bilayers by differential polarized phase fluorometry.Science. 1978 Jun 23;200(4348):1399-401. doi: 10.1126/science.663620. Science. 1978. PMID: 663620 Free PMC article.
-
Time-resolved fluorescence anisotropies of diphenylhexatriene and perylene in solvents and lipid bilayers obtained from multifrequency phase-modulation fluorometry.Biochemistry. 1985 Jan 15;24(2):376-83. doi: 10.1021/bi00323a021. Biochemistry. 1985. PMID: 3978080 Free PMC article.
-
Provisional Peer-Reviewed Toxicity Values for Perylene (CASRN 198-55-0).Cincinnati (OH): U.S. Environmental Protection Agency; 2023 Feb. Cincinnati (OH): U.S. Environmental Protection Agency; 2023 Feb. PMID: 38484101 Free Books & Documents. Review. No abstract available.
Cited by
-
Fluorescence probe partitioning between Lo/Ld phases in lipid membranes.Biochim Biophys Acta. 2007 Sep;1768(9):2182-94. doi: 10.1016/j.bbamem.2007.05.012. Epub 2007 May 21. Biochim Biophys Acta. 2007. PMID: 17588529 Free PMC article.
-
Fluorescence depolarization measurements on oriented membranes.Biophys J. 1988 Jun;53(6):989-1005. doi: 10.1016/S0006-3495(88)83179-5. Biophys J. 1988. PMID: 3165033 Free PMC article.
-
Studies of archaebacterial bipolar tetraether liposomes by perylene fluorescence.Biophys J. 2000 Mar;78(3):1390-9. doi: 10.1016/S0006-3495(00)76692-6. Biophys J. 2000. PMID: 10692324 Free PMC article.
-
Light Quenching and Fluorescence Depolarization of Rhodamine B and Applications of This Phenomenon to Biophysics.J Phys Chem. 1994 Jan;98(1):334-342. doi: 10.1021/j100052a055. J Phys Chem. 1994. PMID: 32454536 Free PMC article.
-
Adenylate cyclase and membrane fluidity. The repressor hypothesis.Mol Cell Biochem. 1984;60(1):17-31. doi: 10.1007/BF00226298. Mol Cell Biochem. 1984. PMID: 6323963 Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
