Flow-mediated endothelial mechanotransduction
- PMID: 7624393
- PMCID: PMC3053532
- DOI: 10.1152/physrev.1995.75.3.519
Flow-mediated endothelial mechanotransduction
Abstract
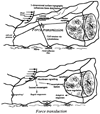
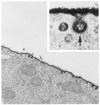
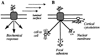
Mechanical forces associated with blood flow play important roles in the acute control of vascular tone, the regulation of arterial structure and remodeling, and the localization of atherosclerotic lesions. Major regulation of the blood vessel responses occurs by the action of hemodynamic shear stresses on the endothelium. The transmission of hemodynamic forces throughout the endothelium and the mechanotransduction mechanisms that lead to biophysical, biochemical, and gene regulatory responses of endothelial cells to hemodynamic shear stresses are reviewed.
Figures






References
-
- Achard C, Aynaud M. Recherches sur l’impregnation histologique de l’endothelium. Arch. Med. Exp. 1907;19:437–450.
-
- Adams DJ, Barakeh J, Laskey R, Van Breemen C. Ion channels and regulation of intracellular calcium in vascular endothelial cells. FASEB J. 1989;3:2389–2400. - PubMed
-
- Aderem A. The MARCKS brothers: a family of protein kinase C substrates. Cell. 1992;71:713–716. - PubMed
-
- Agard DA, Sedat JW. Three-dimensional architecture of a polytene nucleus. Nature Lond. 1983;302:676–681. - PubMed
-
- Ahmad M, Marui N, Alexander RW, Medford RM. Cell type specific transactivation of the VCAM-1 promoter through an NFκB enhancer motif. J. Biol. Chem. In press. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

