The acidic activation domain of the Epstein-Barr virus transcription factor R interacts in vitro with both TBP and TFIIB and is cell-specifically potentiated by a proline-rich region
- PMID: 8389627
- PMCID: PMC6081622
The acidic activation domain of the Epstein-Barr virus transcription factor R interacts in vitro with both TBP and TFIIB and is cell-specifically potentiated by a proline-rich region
Abstract
In cells latently infected with Epstein-Barr virus (EBV), the expression of two viral transactivators, EB1 and R, is responsible for the switch from latency to a productive cycle. R contains a DNA-binding/dimerization domain localized at the N-terminus. The domain required for transcriptional activation is localized at the C-terminus and contains two regions of very different amino acid composition. The first is very rich in prolines, whereas the second is rich in acidic residues and contains two potential alpha-helices. We investigated the activation potential of these subregions when linked to the heterologous Gal4 DNA-binding domain. We found that the acidic region--more precisely, the second putative alpha-helix--is an activating domain. In contrast, the proline-rich region is insufficient by itself for activation but collaborates with the acidic region in a cell-specific manner to make transactivation more efficient. We demonstrated that R interacts in vitro with the basal transcription factors TBP and TFIIB, and that the acidic domain of R mediates these interactions.
Figures
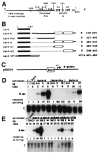
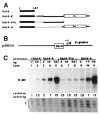
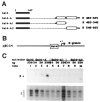

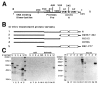
Similar articles
-
Domains of the Epstein-Barr virus (EBV) transcription factor R required for dimerization, DNA binding and activation.Nucleic Acids Res. 1991 May 25;19(10):2661-7. doi: 10.1093/nar/19.10.2661. Nucleic Acids Res. 1991. PMID: 1645863 Free PMC article.
-
Repression by RAZ of Epstein-Barr virus bZIP transcription factor EB1 is dimerization independent.J Gen Virol. 1996 Jul;77 ( Pt 7):1529-36. doi: 10.1099/0022-1317-77-7-1529. J Gen Virol. 1996. PMID: 8757996
-
The Epstein-Barr virus R transactivator (Rta) contains a complex, potent activation domain with properties different from those of VP16.J Virol. 1992 Sep;66(9):5500-8. doi: 10.1128/JVI.66.9.5500-5508.1992. J Virol. 1992. PMID: 1323708 Free PMC article.
-
Epstein-Barr virus (EBV) EB1/Zta protein provided in trans and competent for the activation of productive cycle genes does not activate the BZLF1 gene in the EBV genome.J Gen Virol. 1996 Mar;77 ( Pt 3):501-9. doi: 10.1099/0022-1317-77-3-501. J Gen Virol. 1996. PMID: 8601788
-
Transcriptional activation: enter TFIIB.Trends Biochem Sci. 1991 Sep;16(9):317-8. doi: 10.1016/0968-0004(91)90130-n. Trends Biochem Sci. 1991. PMID: 1949150 Review. No abstract available.
Cited by
-
Ubinuclein, a novel nuclear protein interacting with cellular and viral transcription factors.J Cell Biol. 2000 Mar 20;148(6):1165-76. doi: 10.1083/jcb.148.6.1165. J Cell Biol. 2000. PMID: 10725330 Free PMC article.
-
Interaction of the equine herpesvirus 1 EICP0 protein with the immediate-early (IE) protein, TFIIB, and TBP may mediate the antagonism between the IE and EICP0 proteins.J Virol. 2003 Feb;77(4):2675-85. doi: 10.1128/jvi.77.4.2675-2685.2003. J Virol. 2003. PMID: 12552007 Free PMC article.
-
The nuclear and adherent junction complex component protein ubinuclein negatively regulates the productive cycle of Epstein-Barr virus in epithelial cells.J Virol. 2011 Jan;85(2):784-94. doi: 10.1128/JVI.01397-10. Epub 2010 Nov 17. J Virol. 2011. PMID: 21084479 Free PMC article.
-
The activation domain of herpesvirus saimiri R protein interacts with the TATA-binding protein.J Virol. 1999 Dec;73(12):9756-63. doi: 10.1128/JVI.73.12.9756-9763.1999. J Virol. 1999. PMID: 10559285 Free PMC article.
-
Herpesvirus saimiri open reading frame 50 (Rta) protein reactivates the lytic replication cycle in a persistently infected A549 cell line.J Virol. 2001 Apr;75(8):4008-13. doi: 10.1128/JVI.75.8.4008-4013.2001. J Virol. 2001. PMID: 11264393 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
