17 beta-Estradiol potentiates kainate-induced currents via activation of the cAMP cascade
- PMID: 8642406
- PMCID: PMC6578827
- DOI: 10.1523/JNEUROSCI.16-11-03620.1996
17 beta-Estradiol potentiates kainate-induced currents via activation of the cAMP cascade
Abstract
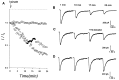
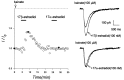
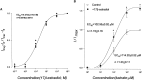
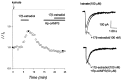
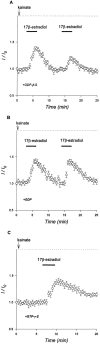
Evidence for nongenomic actions of steroids is now coming from a variety of fields of steroid research. Mechanisms of steroid action are being studied with regard to the membrane receptors and the activation of second messengers. The present study investigated the mechanism for the rapid effect of estrogen on acutely dissociated hippocampal CA1 neurons by using the whole-cell, voltage-clamp recording. Under the perforated patch configuration, 17 beta-estradiol potentiated kainate-induced currents in 38% of tested neurons. The potentiation was stereospecific, rapid in onset, and reversible after the removal of the steroid. Dose-response curves show that the potentiation by 17 beta-estradiol was evident at a concentration as low as 10 nM and saturated at 10 microM. 17 beta-Estradiol did not affect the kinetics (i.e., affinity and cooperativity) and reversal potential of kainate-induced currents. This suggests that the potentiation did not result from direct interaction with kainate receptors nor the activation of ion channels other than kainate receptor-channels. The potentiation by 17 beta-estradiol was similar to the enhancement of kainate-induced currents evoked by 8-bromo-cAMP, and was modulated by an inhibitor of phosphodiesterase (IBMX). The estrogen potentiation was blocked by a specific blocker of PKA (Rp-cAMPS). Under standard recording configuration, the effect was significantly affected by intracellular perfusing with GDP-beta-S or GTP-gamma-S. The data suggest that the potentiation of kainate-induced currents by 17-beta-estradiol was likely a G-protein(s) coupled, cAMP-dependent phosphorylation event. By involvement of this non-genomic mechanism, estrogen may play a role in the modulation of excitatory synaptic transmission in the hippocampus.
Figures



References
-
- Baulieu EE, Robel P. Neurosteroids: a new brain function? J Steroid Biochem Mol Biol. 1990;37:395–403. - PubMed
-
- Bazzett TJ, Bender JB. Sex difference in the rapid and acute effects of estrogen on striatal D2 dopamine receptor binding. Brain Res. 1994;637:163–172. - PubMed
-
- Bergamini CM, Pansini F, Bettocchi S, Segala V, Dallocchio F, Bagni G, Mollica G. Hormonal sensitivity of adenylate cyclase from human endometrium: modulation by estradiol. J Steroid Biochem. 1985;22:299–303. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous
