Cloning and characterization of the signal transduction of four splice variants of the human pituitary adenylate cyclase activating polypeptide receptor. Evidence for dual coupling to adenylate cyclase and phospholipase C
- PMID: 8663363
- PMCID: PMC6721843
- DOI: 10.1074/jbc.271.29.17267
Cloning and characterization of the signal transduction of four splice variants of the human pituitary adenylate cyclase activating polypeptide receptor. Evidence for dual coupling to adenylate cyclase and phospholipase C
Abstract
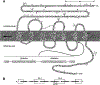
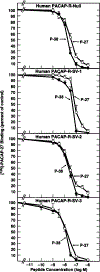
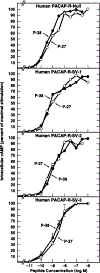
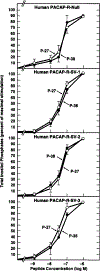
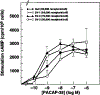
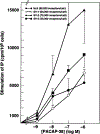
Alternative splicing of two exons of the rat pituitary adenylate cyclase activating polypeptide (PACAP) receptor gene generates four major splice variants that are differentially expressed in specific tissues and variably coupled to intracellular second messengers. To evaluate the potential implications of these findings in human physiology, the human PACAP receptor gene was cloned. Alternative splicing about two exons of the gene allowed for four major splice variants that were subsequently identified on cDNA cloning. Each of the four splice variant cDNAs (null, SV-1, SV-2, and SV-3) was stably expressed in NIH/3T3 cells at similar receptor densities. For each splice variant, PACAP (both PACAP-38 and PACAP-27) had similar affinity and potency for stimulating either adenylate cyclase or phospholipase C. However, each receptor splice variant differed in their ligand-stimulated maximal response (efficacy) for total inositol phosphate accumulation with the SV-2 showing the greatest efficacy, followed by the null, SV-1, and SV-3 splice variants. Therefore, unlike the rat, PACAP binds and stimulates signal transduction with nearly equal affinity and potency for each of the receptor splice variants although with varying efficacy for the stimulation of phospholipase C. These results suggest a novel and potentially important mechanism for a single hormone to not only couple to dual signal transduction cascades but also elicit tissue-specific differential activation of phospholipase C in humans.
Figures
Similar articles
-
Differential signaling and immediate-early gene activation by four splice variants of the human pituitary adenylate cyclase-activating polypeptide receptor (hPACAP-R).Ann N Y Acad Sci. 1996 Dec 26;805:54-64; discussion 64-6. doi: 10.1111/j.1749-6632.1996.tb17473.x. Ann N Y Acad Sci. 1996. PMID: 8993393 Free PMC article.
-
Novel alternatively spliced exon in the extracellular ligand-binding domain of the pituitary adenylate cyclase-activating polypeptide (PACAP) type 1 receptor (PAC1R) selectively increases ligand affinity and alters signal transduction coupling during spermatogenesis.J Biol Chem. 2001 Apr 20;276(16):12938-44. doi: 10.1074/jbc.M009941200. Epub 2001 Jan 29. J Biol Chem. 2001. PMID: 11278585
-
Alternative splicing in the N-terminal extracellular domain of the pituitary adenylate cyclase-activating polypeptide (PACAP) receptor modulates receptor selectivity and relative potencies of PACAP-27 and PACAP-38 in phospholipase C activation.J Biol Chem. 1996 Sep 6;271(36):22146-51. doi: 10.1074/jbc.271.36.22146. J Biol Chem. 1996. PMID: 8703026
-
Differential signal transduction by six splice variants of the pituitary adenylate cyclase-activating peptide (PACAP) receptor.Biochem Soc Trans. 1995 Feb;23(1):133-7. doi: 10.1042/bst0230133. Biochem Soc Trans. 1995. PMID: 7758697 Review. No abstract available.
-
[Pituitary adenylate cyclase-activating polypeptide].Ann Endocrinol (Paris). 1998 Dec;59(5):364-405. Ann Endocrinol (Paris). 1998. PMID: 9949891 Review. French.
Cited by
-
Alternative Splicing of the Pituitary Adenylate Cyclase-Activating Polypeptide Receptor PAC1: Mechanisms of Fine Tuning of Brain Activity.Front Endocrinol (Lausanne). 2013 May 21;4:55. doi: 10.3389/fendo.2013.00055. eCollection 2013. Front Endocrinol (Lausanne). 2013. PMID: 23734144 Free PMC article.
-
Homeodomain protein otp and activity-dependent splicing modulate neuronal adaptation to stress.Neuron. 2012 Jan 26;73(2):279-91. doi: 10.1016/j.neuron.2011.11.019. Neuron. 2012. PMID: 22284183 Free PMC article.
-
PACAP38 and PAC1 receptor blockade: a new target for headache?J Headache Pain. 2018 Aug 7;19(1):64. doi: 10.1186/s10194-018-0893-8. J Headache Pain. 2018. PMID: 30088106 Free PMC article. Review.
-
Agonistic behavior of PACAP6-38 on sensory nerve terminals and cytotrophoblast cells.J Mol Neurosci. 2008 Nov;36(1-3):270-8. doi: 10.1007/s12031-008-9089-z. Epub 2008 Jul 8. J Mol Neurosci. 2008. PMID: 18607779
-
PAC1 regulates receptor tyrosine kinase transactivation in a reactive oxygen species-dependent manner.Peptides. 2019 Oct;120:170017. doi: 10.1016/j.peptides.2018.09.005. Epub 2018 Sep 28. Peptides. 2019. PMID: 30273693 Free PMC article.
References
-
- Miyata A, Arimura A, Dahl RR, Minamino N, Uehara A, Jiang L, Culler MD, and Coy DH (1989) Biochem. Biophys. Res. Commun 164, 567–574 - PubMed
-
- Lam H-C, Takahashi K, Ghatei MA, Kanse SM, Polak JM, and Bloom SR (1990) Eur. J. Biochem 193, 725–729 - PubMed
-
- Shivers BD, Gorcs TJ, Gottschall PE, and Arimura A (1991) Endocrinology 128, 3055–3065 - PubMed
-
- Ishihara T, Shigemoto R, Mori K, Takahashi K, and Nagata S (1992) Neuron 8, 811–819 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

