Tissue specificity of L-pyruvate kinase transgenes results from the combinatorial effect of proximal promoter and distal activator regions
- PMID: 8836739
- PMCID: PMC6138020
Tissue specificity of L-pyruvate kinase transgenes results from the combinatorial effect of proximal promoter and distal activator regions
Abstract
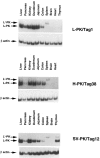
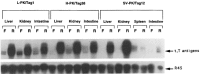
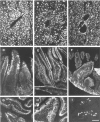
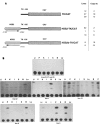
The L-type pyruvate kinase (L-PK) gene is regulated by diet and hormones and expressed at high levels in the hepatocytes, enterocytes, and proximal tubular cells of the kidney and at low levels in the endocrine pancreatic cells. Two regulatory regions have been shown to be important in transgenic mice to confer on a reporter gene a similar tissue-specific and diet-responsive expression: a proximal promoter fragment, with binding sites for the tissue-specific hepatocyte nuclear factors 1 and 4, and presence of the glucose-response element (GIRE) and a distal activator corresponding to a liver-specific hypersensitive site at -3000 bp with respect to the cap site. Although the proximal promoter is able to confer by itself tissue-specific expression on a reporter gene, its activity in vivo is strongly stimulated by the distal activator. To determine the possible role of the distal region on diet responsiveness and tissue specificity of the L-PK gene expression, we have created lines of transgenic mice in which the gene for SV40 T antigen (Tag) was directed by composite regulatory sequences consisting of the L-PK promoter and different enhancers: either the SV40 early enhancer (SV) or the H enhancer of the aldolase A gene (H). The induction of the composite H-PK/Tag and SV-PK/Tag transgenes by a carbohydrate-rich diet in the liver was similar to that of the endogenous L-PK gene. This suggests that in fasted mice the L-PK promoter, and especially the GIRE, is able to silence the activating influence of a strong viral enhancer such as the SV40 enhancer. The H-PK/Tag mice expressed the transgene similarly to the endogenous gene, except in the pancreas, where expression was practically undetectable. Consistently, whereas L-PK/Tag mice develop insulinomas, H-PK/Tag mice develop only hepatomas. In contrast, the transgene expression was partly aberrant in SV-PK/Tag mice. In addition to a normal activation of the transgene in the liver, a strong expression was also detected in the kidney medulla, whereas the transgene was practically silent in enterocytes. Finally, the effect of the distal region (-2070 to -3200) on an ubiquitous promoter was tested by ligating the distal L-PK gene fragment in front of a thymidine kinase/CAT transgene. Such a transgene was constantly expressed in the pancreas and, strikingly, in the brain. It appears, therefore, that the L-PK distal activator exhibits, by itself, a certain neuropancreatic specificity required in combination with the proximal promoter for L-PK gene expression in pancreas endocrine cells.
Figures



References
-
- Baumhueter S.; Mendel D. B.; Conley P. B.; Kuo C. J.; Turk C.; Graves M. K.; Edwards C. A.; Courtois G.; Crabtree G. R. HNF-1 shares three sequence motifs with the POU domain proteins and is identical to LFB1 and APF. Genes Dev. 4:372–379; 1990. - PubMed
-
- Boquet D.; Vaulont S.; Tremp G. L.; Ripoche M.-A.; Daegelen D.; Jami J.; Kahn A.; Raymondjean M. DNaseI hypersensitivity analysis of the L-type pyruvate kinase gene in rats and transgenic mice. Eur. J. Biochem. 207:13–21; 1992. - PubMed
-
- Briegel K.; Lim K. C.; Plank C.; Beug H.; Engel J. D.; Zenke M. Ectopic expression of a conditional GATA-2/estrogen receptor chimera arrests erythroid differentiation in a hormone-dependent manner. Genes Dev. 7:1097–1109; 1993. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous
