Cultured melanocytes from dilute mutant mice exhibit dendritic morphology and altered melanosome distribution
- PMID: 8962090
- PMCID: PMC26171
- DOI: 10.1073/pnas.93.25.14554
Cultured melanocytes from dilute mutant mice exhibit dendritic morphology and altered melanosome distribution
Abstract
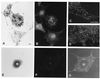
Mutant alleles at the dilute unconventional myosin heavy chain locus cause diluted coat color, opisthotonic seizures, and death. The dilute coat color phenotype is caused by irregular clumping of pigment in the hair, but amounts of melanin are unchanged from wild-type controls. The melanocyte phenotype has been described as adendritic, since hair bulb and Harderian gland melanocytes appear to be rounded in tissue sections. These observations do not exclude the possibility that the processes lack pigment, since the melanocyte shape was judged by the distribution of melanin. We have tested this hypothesis by culturing primary melanocytes from dilute mutant and wild-type mice. The mutant melanocytes do not lack processes; instead, they exhibit a concentrated perinuclear distribution of melanosomes, while wild-type melanocytes have a very uniform cytoplasmic distribution of melanosomes. Electron micrographs show no detectable differences in melanosome morphology or maturation between dilute and wild-type melanocytes. Immunofluorescence experiments indicate that the dilute protein is concentrated in regions of the cytoplasm that contain melanosomes. These experiments show that the dilute myosin is necessary for the localization of melanosomes, either by active transport or tethering.
Figures



References
Publication types
MeSH terms
Substances
Associated data
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

